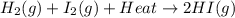The reaction below shows a system in equilibrium. how would a decrease in temperature affect this reaction? a. the rate of formation of the gases would increase. b. the equilibrium of the reaction would shift to the left. c. the equilibrium would shift to cause the gases to sublime into solids. d. the chemicals on the left would quickly form the chemical on the right.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 00:00
The pressure inside a hydrogen-filled container was 2.10 atm at 21 ? c. what would the pressure be if the container was heated to 92 ? c ?
Answers: 2



Chemistry, 23.06.2019 05:00
What is dhmo? hint: you find it everywhere something is wet..
Answers: 1
You know the right answer?
The reaction below shows a system in equilibrium. how would a decrease in temperature affect this r...
Questions

Mathematics, 26.08.2019 23:00

Social Studies, 26.08.2019 23:00


History, 26.08.2019 23:00


Biology, 26.08.2019 23:00


Computers and Technology, 26.08.2019 23:00

Mathematics, 26.08.2019 23:00



Mathematics, 26.08.2019 23:00