
Chemistry, 23.06.2019 03:40 ElegantEmerald
The following questions a24 - a26 relate to 100 ml of 0.0150 m solution of benzoic acid (c6h3cooh). ka(c6h3cooh) = 6.4 x 10^-5. what is the ph of the solution after the addition of 1 x 10^-3 moles of naoh? you may assume no volume change to the solution upon addition of the naoh.

Answers: 2
Another question on Chemistry


Chemistry, 21.06.2019 14:00
Perform the following mathematical operations and report the answer to the appropriate number of significant figures 5.87998 + 3.100
Answers: 2

Chemistry, 22.06.2019 13:30
An animal cell loses the ability to convert energy stored in food to energy that the cell can use. which of the cell's organelles has stopped working? a.the mitochondria b.the nucleus c.the vacuoles d.the endoplasmic reticulum
Answers: 1

You know the right answer?
The following questions a24 - a26 relate to 100 ml of 0.0150 m solution of benzoic acid (c6h3cooh)....
Questions


Chemistry, 12.04.2021 21:20

Mathematics, 12.04.2021 21:20







Computers and Technology, 12.04.2021 21:20



Mathematics, 12.04.2021 21:20


Mathematics, 12.04.2021 21:20

Physics, 12.04.2021 21:20



Mathematics, 12.04.2021 21:20

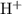 be
be 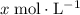 . Note that
. Note that 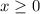 .
.
![\displaystyle \frac{[\mathrm{C_6H_5COO^{-}}]\cdot [\mathrm{H^{+}}]}{[\mathrm{C_6H_5COOH}]} = \mathrm{pK}_{a}](/tpl/images/0006/5046/265ec.png) .
.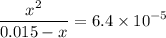 .
. :
: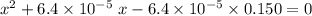 .
.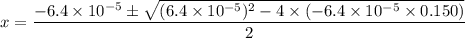 .
.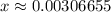 .
.![\rm [H^{+}] = 0.00306655\; mol\cdot L^{-1}](/tpl/images/0006/5046/db736.png) .
.![\displaystyle \mathrm{pH} = -\log_{10}{[\mathrm{H^{+}}]} = 2.513](/tpl/images/0006/5046/141cf.png) .
.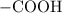 . Benzoic acid is thus a monoprotic acid. Each mole of the acid will react with only one mole of
. Benzoic acid is thus a monoprotic acid. Each mole of the acid will react with only one mole of 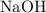 . The 100 mL solution initially contains
. The 100 mL solution initially contains 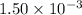 moles of benzoic acid. The
moles of benzoic acid. The 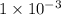 moles of
moles of 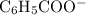 (from the salt
(from the salt 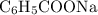 ) and
) and 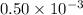 moles of
moles of 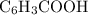 .
.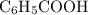 and the conjugate base of the acid
and the conjugate base of the acid 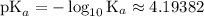 for benzoic acid.
for benzoic acid.![\begin{aligned}\mathrm{pH} &= \mathrm{pK}_{a} + \log{\frac{{[\text{Conjugate Base}]}}{[\text{Weak Acid}]}} \\ &= \mathrm{pK}_{a} + \log{\frac{{[\mathrm{C_6H_5COO^{-}}]}}{[\mathrm{C_6H_5COOH}]}}\\ &= 4.19382 + \log{\frac{0.01}{0.005}}\\ &\approx 4.495 \end{aligned}](/tpl/images/0006/5046/20b2b.png) .
.


