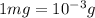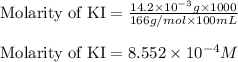Chemistry, 22.06.2019 11:50 hamidaakter936848
Calculate the molarity of each of the following solutions. part a) 0.12 mol of lino3 in 5.5 l of solution part b) 60.7 g c2h6o in 2.48 l of solution part c) 14.2 mg ki in 100 ml of solution

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 23:00
What is the formula that this ionic compounds could form sr2+p3-o2-
Answers: 3

Chemistry, 22.06.2019 01:30
Phosphorous acid, h3po3(aq) , is a diprotic oxyacid that is an important compound in industry and agriculture. the values of phosphorous acid are 1.30 6.70 calculate the ph for each of the given points in the titration of 50.0 ml of 1.5 m h3po3(aq) with 1.5 m koh(aq) .
Answers: 3

Chemistry, 22.06.2019 05:00
What forms when chemical reactions combine pollution with sunlight?
Answers: 1

You know the right answer?
Calculate the molarity of each of the following solutions. part a) 0.12 mol of lino3 in 5.5 l of sol...
Questions

History, 19.10.2019 03:00

Mathematics, 19.10.2019 03:00

English, 19.10.2019 03:00



History, 19.10.2019 03:00

Social Studies, 19.10.2019 03:00

Health, 19.10.2019 03:00

History, 19.10.2019 03:00


Geography, 19.10.2019 03:00

Geography, 19.10.2019 03:00






Health, 19.10.2019 03:00


Mathematics, 19.10.2019 03:00

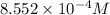
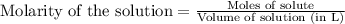
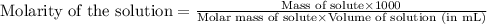
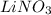 in 5.5 L of solution
in 5.5 L of solution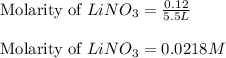
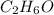 in 2.48 L of solution
in 2.48 L of solution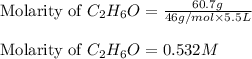
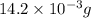 (Conversion factor:
(Conversion factor: