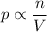Chemistry, 10.07.2019 23:30 demotte79991
5. assuming the temperature is held constant, how could you increase the pressure inside a
container by a factor of 37

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 14:50
Consider the following multistep reaction: a b→ab(slow) a ab→a2b(fast)−−−−−−−−−−−−−−−−− 2a b→a2b(overall) based on this mechanism, determine the rate law for the overall reaction. express your answer in standard masteringchemistry format. for example, if the rate law is k[a]3[b]2 type k*[a]^3*[b]^2
Answers: 3

Chemistry, 22.06.2019 17:30
Air can be considered a mixture. which statement does not explain why?
Answers: 1

Chemistry, 22.06.2019 23:00
What is the solubility-product constant of barium sulfate, baso4, if a saturated solution is 1.03 ´ 10-5 m?
Answers: 3
You know the right answer?
5. assuming the temperature is held constant, how could you increase the pressure inside a
con...
con...
Questions


Mathematics, 28.10.2019 02:31




English, 28.10.2019 02:31


Mathematics, 28.10.2019 02:31

Mathematics, 28.10.2019 02:31

Mathematics, 28.10.2019 02:31





French, 28.10.2019 02:31

Biology, 28.10.2019 02:31



English, 28.10.2019 02:31