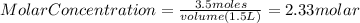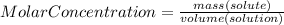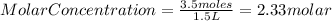Chemistry, 12.07.2019 04:10 Wolfgirl2032
Astudent adds 3.5 moles of solute to enough water to make a 1500ml solution. what is the concentration?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 02:00
The alkali metals (group 1) consist of lithium (3), sodium (11), potassium (19), rubidium (37), cesium (55), and francium (87). they are soft, metallic solids with low densities and low melting points. based on the data shown in figure 1, how many valence electrons do alkali metals share?
Answers: 3

Chemistry, 22.06.2019 10:50
8) a mixture of he, ne and ar has a pressure of 7.85 atm. if the ne has a mole fraction of 0.47 and 8) ar has a mole fraction of 0.23, what is the pressure of he? a) 4.2 atm b) 3.7 atm c) 5.5 atm d) 2.4 atm e) 1.8 atm
Answers: 1

Chemistry, 22.06.2019 14:50
Consider the following multistep reaction: a b→ab(slow) a ab→a2b(fast)−−−−−−−−−−−−−−−−− 2a b→a2b(overall) based on this mechanism, determine the rate law for the overall reaction. express your answer in standard masteringchemistry format. for example, if the rate law is k[a]3[b]2 type k*[a]^3*[b]^2
Answers: 3

Chemistry, 22.06.2019 19:10
Δu of , in kj/kg, as it isto k, (a)as a of , (b) at , (c) at .
Answers: 2
You know the right answer?
Astudent adds 3.5 moles of solute to enough water to make a 1500ml solution. what is the concentrati...
Questions




Computers and Technology, 14.09.2019 05:30


Computers and Technology, 14.09.2019 05:30


Computers and Technology, 14.09.2019 05:30


Computers and Technology, 14.09.2019 05:30





Computers and Technology, 14.09.2019 05:30

Computers and Technology, 14.09.2019 05:30