
Chemistry, 14.07.2019 18:10 Natavia3402
Find the enthalpy of neutralization of hcl and naoh. 137 cm3 of 2.6 mol dm-3 hydrochloric acid was neutralized by 137 cm3 of 2.6 mol dm-3 naoh. the temperature rose from 298 k to 325.8 k. the specific heat capacity is the same as water, 4.18 j/k g.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 22:30
What is the work done by the electric force to move a 1 c charge from a to b?
Answers: 2

Chemistry, 23.06.2019 01:00
What type of chemical bond is formed between two atoms of bromine 1. metallic 2. hydrogen 3. ionic 4. covalent
Answers: 1

Chemistry, 23.06.2019 07:10
1) a light bulb takes in 30 of energy per second. it transfers 3j as use energy. calculate the efficiency. second. it transfers 3j as useful light energy and 27j as heat energy. calculate the efficiency
Answers: 1

You know the right answer?
Find the enthalpy of neutralization of hcl and naoh. 137 cm3 of 2.6 mol dm-3 hydrochloric acid was n...
Questions


Mathematics, 30.11.2019 14:31


History, 30.11.2019 14:31



Mathematics, 30.11.2019 14:31


Mathematics, 30.11.2019 14:31

Health, 30.11.2019 14:31





Biology, 30.11.2019 14:31


English, 30.11.2019 14:31


History, 30.11.2019 14:31

History, 30.11.2019 14:31

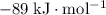 .
.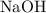 formula units will neutralize one mole of
formula units will neutralize one mole of 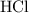 to produce one mole of water.
to produce one mole of water. 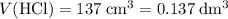 .
.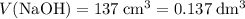 .
.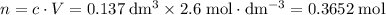 .
.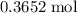 of
of 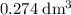 . Assume that the density of the solution is equal to that of water under room temperature.
. Assume that the density of the solution is equal to that of water under room temperature. 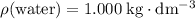 . The mass of the liquid will be
. The mass of the liquid will be 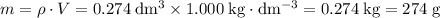 .
.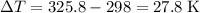 .
.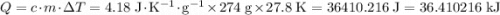 .
. .
.

