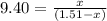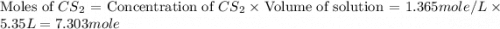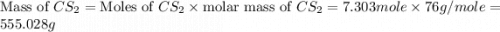Chemistry, 24.07.2019 22:20 nikejose11
Carbon disulfide is prepared by heating sulfur and charcoal. the chemical equation is s2(g)+c(s)↽−−⇀cs2(=9.40 at 900 k how many grams of cs2(g) can be prepared by heating 8.08 mol s2(g) with excess carbon in a 5.35 l reaction vessel held at 900 k until equilibrium is attained?

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 16:40
Who is better, messi or cristiano, i need this for a chemistry class. asap
Answers: 1

Chemistry, 22.06.2019 22:30
Gusing the milligrams of ascorbic acid you entered above, the ratio of total sample volume to aliquot volume, and the total milligrams of the vitamin c tablet that you dissolved, calculate the mass of ascorbic acid in the vitamin c tablet for each trial. do this by scaling up to find the amount (mg) of ascorbic acid in your 250 ml flask. enter your calculated mass of ascorbic acid in the vitamin c tablet, for each trial. be sure to enter your calculated mass in the corresponding order that you entered your milligrams of ascorbic acid. the milligrams of ascorbic acid you entered for entry #1 previously should correspond to the mass of ascorbic acid that you enter for entry #1 here.
Answers: 1

Chemistry, 23.06.2019 01:00
The primary products of complete combustion of fossil fuels are a. carbon dioxide and water b. methane and water c. carbon monoxide and water d. carbon dioxide and carbon monoxide
Answers: 1

Chemistry, 23.06.2019 06:30
Aplanet similar to earth has four moons roughly the same distance away. the moon that will most affect tides on the planet is the one that has the greatest a) mass. b) volume. c) density. d) amount of water.
Answers: 1
You know the right answer?
Carbon disulfide is prepared by heating sulfur and charcoal. the chemical equation is s2(g)+c(s)↽−−⇀...
Questions


Mathematics, 26.10.2019 21:43

Mathematics, 26.10.2019 21:43



Mathematics, 26.10.2019 21:43

Biology, 26.10.2019 21:43

Biology, 26.10.2019 21:43



English, 26.10.2019 21:43


Mathematics, 26.10.2019 21:43


Mathematics, 26.10.2019 21:43



Social Studies, 26.10.2019 21:43

Biology, 26.10.2019 21:43

Mathematics, 26.10.2019 21:43

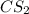 is, 555.028 grams
is, 555.028 grams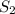 .
.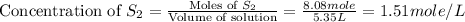
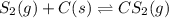
![K_c=\frac{[CS_2]}{[S_2]}](/tpl/images/0128/8287/0a94f.png)