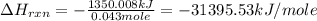Chemistry, 25.07.2019 18:10 tateandvioletAHS14AY
The combustion of titanium with oxygen produces titanium dioxide: ti (s) + o2 (g) → tio2 (s) when 2.060 g of titanium is combusted in a bomb calorimeter, the temperature of the calorimeter increases from 25.00°c to 91.60°c. in a separate experiment, the heat capacity of the calorimeter is measured to be 9.84 kj/k. the heat of reaction for the combustion of a mole of ti in this calorimeter is kj/mol.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 00:00
Explain which group an element with the electron configuration 1s2 2s2 2p6 3s2 3p6 3d1 4s2 belongs to.
Answers: 3

Chemistry, 22.06.2019 05:50
Astudent made a graph plotting the progress of a reaction over time. the student forgot to label the y-axis of the graph. a graph is shown with two graph lines. one graph line starts at a higher position on the y axis and slopes downwards towards the right. the other graph line starts at a lower position on the y axis and slopes upwards towards the right. the two graph lines stop short of intersecting each other and continue as separate lines which gradually become straight and parallel to the x axis. a vertical line is shown at a point where the two graph lines finally became parallel to the x axis. this vertical line is labeled equilibrium. the title on the x axis is time and an arrow pointing towards the right is shown above time. the title on the y axis is left blank. what best explains the label that the student should use on the y-axis? amount, because as the amount of product decreases, the amount of reactant increases over time. reaction rate, because forward and backward reaction become equal at equilibrium. amount, because the amounts of reactants and products become constant after equilibrium is reached. reaction rate, as the rate of forward reaction increases and rate of backward reaction decreases over time.
Answers: 3

Chemistry, 22.06.2019 11:40
Which of these expressions are correct variations of the combined gas law? p1v1t2 = p2v2t1 both
Answers: 2

Chemistry, 23.06.2019 00:00
What is the empirical formula of a compound that is 50.7% antimony and 49.3% selenium ?
Answers: 2
You know the right answer?
The combustion of titanium with oxygen produces titanium dioxide: ti (s) + o2 (g) → tio2 (s) when 2...
Questions

English, 24.09.2019 10:30


Mathematics, 24.09.2019 10:30

Mathematics, 24.09.2019 10:30

Mathematics, 24.09.2019 10:30


Business, 24.09.2019 10:30


History, 24.09.2019 10:30


Chemistry, 24.09.2019 10:30


Mathematics, 24.09.2019 10:30


Biology, 24.09.2019 10:30




History, 24.09.2019 10:30

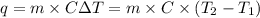
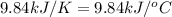
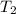 = final temperature =
= final temperature = 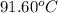
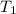 = initial temperature =
= initial temperature = 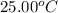
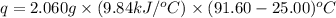
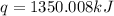
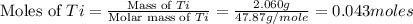
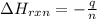
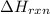 = enthalpy of reaction = ?
= enthalpy of reaction = ?