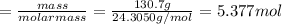Chemistry, 30.07.2019 04:10 luvpeaceandsocc6312
Assuming an efficiency of 49.50%, calculate the actual yield of magnesium nitrate formed from 130.7 g of magnesium and excess copper(ii) nitrate. mg+cu(no3)2⟶mg(no3)2+cu actual yield:

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 14:30
For the reaction shown, find the limiting reactant for each of the following initial amounts of reactants. 4al(s)+3o2(g)→2al2o3(s) a) 1 molal, 1 mol o2 b) 4 molal, 2.6 mol o2 c) 16 molal, 13 mol o2 d) 7.4 molal, 6.5 mol o2
Answers: 3

Chemistry, 22.06.2019 17:30
Observation and experimentation have led many scientists to accept a theory about the origin of the universe. this theory is called the big bang theory. scientific evidence collected and observed by scientists around the world suggests that the universe is ever expanding from a hot and dense initial state. what makes this a scientific theory? (2 points)
Answers: 2

Chemistry, 23.06.2019 10:30
Ireally need ! calcium metal reacts with a potassium chloride solution to form calcium chloride and potassium ions. balance this reaction. (s) + (aq) → cacl2(s) + +(aq) a) 1, 2, 1, 2 b) 1, 2, 1, 1 c) 1, 1, 1, 1 d) 2, 1, 2, 1
Answers: 1

Chemistry, 23.06.2019 11:20
The chemical composition of soil varies with depth. an article in communications in soil science and plant analysis describes chemical analyses of soil taken from a farm in western australia. fifty specimens were each taken at depths 50 and 250 cm. at a depth of 50 cm, the average no3 concentration (in mg/l) was 88.5 with a standard deviation of 49.4. at a depth of 250 cm, the average concentration was 110.6 with a standard deviation of 51.5. find a 95% confidence interval for the difference in no3 concentrations at the two depths.
Answers: 1
You know the right answer?
Assuming an efficiency of 49.50%, calculate the actual yield of magnesium nitrate formed from 130.7...
Questions

Mathematics, 02.04.2020 19:35





Mathematics, 02.04.2020 19:35


Social Studies, 02.04.2020 19:35



Mathematics, 02.04.2020 19:35

History, 02.04.2020 19:35



English, 02.04.2020 19:35

Mathematics, 02.04.2020 19:35


Chemistry, 02.04.2020 19:35

History, 02.04.2020 19:35