
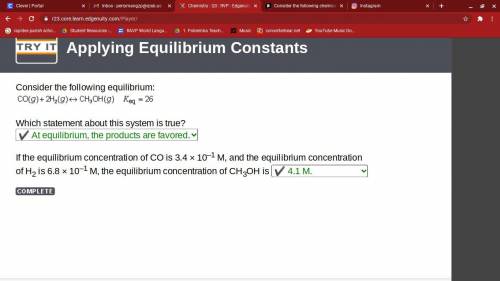
Consider the following chemical reaction: co (g) + 2h2(g) ↔ ch3oh(g) at equilibrium in a particular experiment, the concentrations of co and h2 were 0.15 m and0.36 m, respectively. what is the equilibrium concentration of ch3oh? the value of keq for this reaction is 14.5 at the temperature of the experiment. what is the equilibrium concentration of ch3oh?
a) 14.5b) 7.61 x 10 ^ -3c) 2.82 x 10 ^ -1d) 3.72 x 10 ^ -3e) 1.34 x 10 ^ -3

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 17:50
Which statement is a reason to support population regulation? a) it is unethical for us to control birth control rates b) humans have the freedom to produce as many children as desired c) the gap between the rich and poor has been narrowing since 1960 d) billions more people on the earth will intensify many environmental and social problems
Answers: 1

Chemistry, 21.06.2019 20:40
Astudent made the lewis dot diagram of a compound shown. what is the error in the lewis dot diagram? a)an o atom should transfer all of its six electrons to mg because the formula is mgo b) both electrons of mg should be transferred to one o adam because the formula is mgo c) the electrons should be transferred from each o add him to capital mg because mg has fewer electrons d) the number of dots around mg should be four because it has to transfer two electrons to each o
Answers: 1

Chemistry, 22.06.2019 05:30
What royal scientist used the 29th day of frozen vapor to encounter elements for mastering new culinary creations?
Answers: 1

Chemistry, 22.06.2019 06:40
Which statement is usually true about the relationship between activation energy and reaction rates? low activation energy barriers result in low rates. high activation energy barriers result in low rates. low activation energy barriers result in no reaction. high activation energy barriers result in no reaction.
Answers: 3
You know the right answer?
Consider the following chemical reaction: co (g) + 2h2(g) ↔ ch3oh(g) at equilibrium in a particular...
Questions


Mathematics, 17.04.2021 22:10


Mathematics, 17.04.2021 22:10

Mathematics, 17.04.2021 22:10


English, 17.04.2021 22:10


English, 17.04.2021 22:10

Mathematics, 17.04.2021 22:10

Mathematics, 17.04.2021 22:10

Computers and Technology, 17.04.2021 22:10


Business, 17.04.2021 22:10

Social Studies, 17.04.2021 22:10

Social Studies, 17.04.2021 22:10




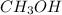 will be, (C)
will be, (C) 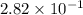
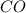 at equilibrium = 0.15 M
at equilibrium = 0.15 M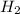 at equilibrium = 0.36 M
at equilibrium = 0.36 M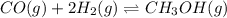
![K_c=\frac{[CH_3OH]}{[CO][H_2]^2}](/tpl/images/0155/4298/4cf94.png)
![14.5=\frac{[CH_3OH]}{(0.15)\times (0.36)^2}](/tpl/images/0155/4298/2d443.png)
![[CH_3OH]=2.82\times 10^{-1}M](/tpl/images/0155/4298/5b3a4.png)



