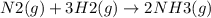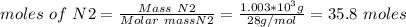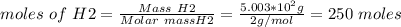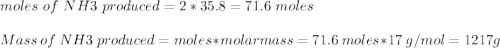Chemistry, 31.07.2019 19:10 zdwilliams1308
Ammonia is produced from the reaction of nitrogen and hydrogen according to the following balanced equation: n2 1 g 2 1 3h2 1 g 2 h 2nh3 1 g 2
a. what is the maximum mass of ammonia that can be produced from a mixture of 1.00 3 103 g n2 and 5.00 3 102 g h2

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 04:20
Which formula can be used to calculate the molar mass of ammonia (nh3)? molar mass of n + molar mass of h 3 × molar mass of n + molar mass of h molar mass of n + 3 × molar mass of h 3 × molar mass of n + 3 × molar mass of h
Answers: 1


Chemistry, 22.06.2019 10:30
What is the empirical formula of c6h18o3? ch3o c2h5o c2h6o c2h5o5
Answers: 1

Chemistry, 22.06.2019 16:00
Which process transfers heat from inside earth to its surface? convection currents in mantle pulling away of tectonic plates drawing in of tectonic plates convection currents in crust
Answers: 1
You know the right answer?
Ammonia is produced from the reaction of nitrogen and hydrogen according to the following balanced e...
Questions


Arts, 16.10.2020 23:01

English, 16.10.2020 23:01



Geography, 16.10.2020 23:01

Arts, 16.10.2020 23:01


Mathematics, 16.10.2020 23:01


Mathematics, 16.10.2020 23:01

Mathematics, 16.10.2020 23:01

Mathematics, 16.10.2020 23:01


Mathematics, 16.10.2020 23:01

Mathematics, 16.10.2020 23:01


English, 16.10.2020 23:01


History, 16.10.2020 23:01