
2h2 + 2 no → n2 + 2 h2o the observed rate expression, under some conditions, is: rate = k[no]2 which of the following mechanisms are consistent with these data? select all that are true. step 1 h2 + no → h2o + n (slow) step 2 n + no → n2 + o (fast) step 3 o + h2 → h2o (fast)

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 19:10
Which statement correctly describes the phosphate ion, ? it is composed of one phosphorus atom and four oxygen atoms covalently bonded together, and there is a –3 charge distributed over the entire ion. it is composed of one phosphorus atom and four oxygen atoms covalently bonded together, and there is a –3 charge on the phosphorus atom. it is composed of one phosphorus atom and four oxygen atoms ionically bonded together, and there is a –3 charge distributed over the entire ion. it is composed of one phosphorus atom and four oxygen atoms ionically bonded together, and there is a –3 charge on the phosphorus atom.
Answers: 3

Chemistry, 23.06.2019 05:00
Match each term to its description. match term definition excess reactant a) reactant that can produce a lesser amount of the product limiting reactant b) amount of product predicted to be produced by the given reactants theoretical yield c) reactant that can produce more of the product
Answers: 3

Chemistry, 23.06.2019 05:50
What is the molecular formula of ferrous nitrate and ferric nitrate
Answers: 2
You know the right answer?
2h2 + 2 no → n2 + 2 h2o the observed rate expression, under some conditions, is: rate = k[no]2 whic...
Questions




















Computers and Technology, 27.08.2019 20:20

![Rate \propto [\text{concentration of reactant}]^{n}](/tpl/images/0164/6015/0f61b.png)
![[\text{concentration of reactant}]^{n}](/tpl/images/0164/6015/2c03d.png)
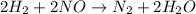
![k[H_{2}][NO]](/tpl/images/0164/6015/98c97.png)
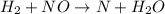 (slow)
(slow)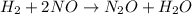 (slow)
(slow)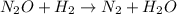 (fast)
(fast)

