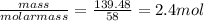Chemistry, 05.08.2019 22:30 izzynikkie
Consider the thermochemical equation for the combustion of acetone (c3h6o), the main ingredient in nail polish remover: c3h6o(l)+4 o2(g)→3 co2(g)+3 h2o(g)δh°rxn=−1790 kj if a bottle of nail polish remover contains 177 ml of acetone, how much heat is released by its complete combustion? the density of acetone is 0.788 g/ml.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 03:40
Astudent is given a sample of a blue copper sulfate hydrate. he weighs the sample in a dry covered porcelain crucible and got a mass of 23.875 g for the crucible, lid, and sample. the mass of the empty crucible and lid was found earlier to be 22.652 g. he then heats the crucible to expel the water of hydration, keeping the crucible at red heat for 10 minutes with the lid slightly ajar. on colling, he finds the mass of crucible, lid, and contents to be 23.403 g. the sample was changed in the process to very light clue anhydrous cuso4. if there are again 100.0 g of hydrate, how many grams of cuso4 are in it? how many moles of cuso4? (hint: molar mass of cuso4 = 159.6 g / mole. what per cent of the hydrate is cuso4? you may convert the mass of cuso4 to moles.)
Answers: 3

Chemistry, 22.06.2019 11:00
As air becomes more dense, (select all that apply) o. air weighs less o. gas molecules are closer together o. air is colder o. air weighs more o. gas molecules are further apart o. air is hotter
Answers: 3

Chemistry, 23.06.2019 00:00
Total the mass on the syringe. record it in the correct row of the data table. kg done click and drag weights to change the pressure. click the syringe to zoom in and see the volume. intro
Answers: 3

Chemistry, 23.06.2019 01:10
A5.00 g of a in . g of at aa 5.00 g of b in . g of .?at .
Answers: 1
You know the right answer?
Consider the thermochemical equation for the combustion of acetone (c3h6o), the main ingredient in n...
Questions


Computers and Technology, 12.10.2019 09:30

History, 12.10.2019 09:30

English, 12.10.2019 09:30


Mathematics, 12.10.2019 09:30


Chemistry, 12.10.2019 09:30


History, 12.10.2019 09:30

English, 12.10.2019 09:30

Biology, 12.10.2019 09:30

Biology, 12.10.2019 09:30

Mathematics, 12.10.2019 09:30




History, 12.10.2019 09:30

Mathematics, 12.10.2019 09:30