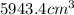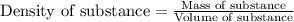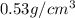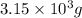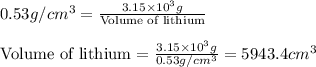Chemistry, 12.08.2019 22:20 maddireigh6403
Lithium is the least dense metal known (density =0.53 g/cm^3). what is the volume occupied by 3.15 x 10^3 g of lithium?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 14:50
Consider the following multistep reaction: a b→ab(slow) a ab→a2b(fast)−−−−−−−−−−−−−−−−− 2a b→a2b(overall) based on this mechanism, determine the rate law for the overall reaction. express your answer in standard masteringchemistry format. for example, if the rate law is k[a]3[b]2 type k*[a]^3*[b]^2
Answers: 3

Chemistry, 22.06.2019 19:20
For a research project, a student decided to test the effect of the lead(ii) ion (pb2+) on the ability of salmon eggs to hatch. this ion was obtainable from the water‐soluble salt, lead(ii) nitrate, which the student decided to make by the following reaction. pbo(s) + 2 hno3(aq) → pb(no3)2(aq) + h2o losses of product for various reasons were expected, and a yield of 86.0% was expected. in order to have 5.00 g of product at this yield, how many grams of pbo should be reacted? (assume that sufficient nitric acid, hno3, would be used.)
Answers: 1

Chemistry, 22.06.2019 20:00
Nitrogen dioxide decomposes according to the reaction 2 no2(g) ⇌ 2 no(g) + o2(g) where kp = 4.48 × 10−13 at a certain temperature. if 0.70 atm of no2 is added to a container and allowed to come to equilibrium, what are the equilibrium partial pressures of no(g) and o2(g)
Answers: 2

Chemistry, 23.06.2019 00:00
How many moles of water are created if 3 moles of hydrogen react completely with excess oxygen?
Answers: 3
You know the right answer?
Lithium is the least dense metal known (density =0.53 g/cm^3). what is the volume occupied by 3.15 x...
Questions


Mathematics, 03.02.2021 20:20

Mathematics, 03.02.2021 20:20

Arts, 03.02.2021 20:20

Mathematics, 03.02.2021 20:20

Mathematics, 03.02.2021 20:20

Chemistry, 03.02.2021 20:20

English, 03.02.2021 20:20

Mathematics, 03.02.2021 20:20


Mathematics, 03.02.2021 20:20

Computers and Technology, 03.02.2021 20:20

Mathematics, 03.02.2021 20:20


Mathematics, 03.02.2021 20:20

Geography, 03.02.2021 20:20