
Chemistry, 13.08.2019 04:30 maddylosin1032
Cl−(aq)+no−3(aq) → no(g)+cl2(g) express your answer as a chemical equation. identify all of the phases in your answer.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 03:30
In this chemical reaction, 325 grams of barium (ba) react completely. how many moles of lithium (li) are produced?
Answers: 1

Chemistry, 22.06.2019 18:00
Chlorophyll a had the molecular formula c55h72mgn4o5 how many atoms are in this molecule
Answers: 2

Chemistry, 23.06.2019 04:20
The equation below shows the reaction of zinc with hydrochloric acid (hcl). zn (s) + 2 hcl (aq) —> zncl2 (aq) + h2 (g) what will happen if the concentration of hcl is decreased? a. more zncl2 will be produced. b. the reaction rate will slow down. c. the hydrochloric acid will become more acidic. d. the reaction will produce water instead of hydrogen gas.
Answers: 1

Chemistry, 23.06.2019 12:50
Acertain reaction has a activation energy of 54.0 kj/mol. as the temperature is increased from 22c to a higher temperature, the rate constant increases by a factor of 7.00. calculate the higher temperature. c (report only numerical answer)
Answers: 3
You know the right answer?
Cl−(aq)+no−3(aq) → no(g)+cl2(g) express your answer as a chemical equation. identify all of the phas...
Questions






Mathematics, 29.07.2019 19:00


English, 29.07.2019 19:00

English, 29.07.2019 19:00

History, 29.07.2019 19:00




Spanish, 29.07.2019 19:00




Geography, 29.07.2019 19:00

Biology, 29.07.2019 19:00

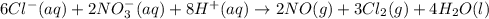
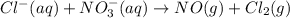
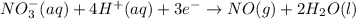 ......(1)
......(1)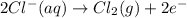 .......(2)
.......(2)

