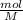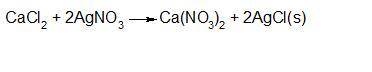Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 23:00
Ahypothrticalax type of ceramic material is known to have a density of 2.10 g/cm3 and a unit cell of cubic symmetry with a cell edge length of 0.57 nm. the atomic weights of the a and x elements are 28.5and 30.0 g/mol, respectively. on the basis of this information, which of the following crystal structures is (are) possible for this material: sodium chloride, cesium chloride, or zinc blende
Answers: 1


Chemistry, 22.06.2019 03:40
Kc = 0.040 for the system below at 450oc. if a reaction is initiated with 0.40 mole of cl2 and 0.40 mole of pcl3 in a 2.0 liter container, what is the equilibrium concentration of cl2 in the same system? pcl5(g) ⇄ pcl3(g) + cl2(g)
Answers: 3

Chemistry, 22.06.2019 06:30
Summarize possible ways in which phases of matter could combine to form a solution.
Answers: 2
You know the right answer?
Consider that you have 330 ml of a 0.37 m aqueous solution of calcium chloride. what volume (in ml)...
Questions

Social Studies, 30.07.2019 06:30


Business, 30.07.2019 06:30






Chemistry, 30.07.2019 06:30

English, 30.07.2019 06:30

Biology, 30.07.2019 06:30

Mathematics, 30.07.2019 06:30






Mathematics, 30.07.2019 06:30


Biology, 30.07.2019 06:30