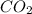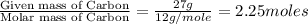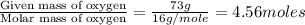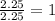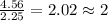Chemistry, 19.08.2019 22:10 lindsey0456
What is the empirical formula of an unknown compound if was determined by a laboratory experiment that it is made up of 27% by mass of carbon and 73% by mass of oxygen?

Answers: 1
Another question on Chemistry

Chemistry, 23.06.2019 04:31
What are the coefficients that will balance the skeleton equation below? n2 + h2 → nh3
Answers: 1

Chemistry, 23.06.2019 06:30
The polarity of an oxygen-hydrogen bond is higher than the polarity of a nitrogen-hydrogen bond, allowing amines to be more soluble than alcohols.
Answers: 3

Chemistry, 23.06.2019 12:30
D5w is shorthand for a 5% glucose rehydration fluid used in ivs. the doctor orders 1200 ml d5w@ 30 gtts/min. you have an iv tube that delivers 18 gtts/cc. how many hours will it take for the 1200 cc bottle to infuse?
Answers: 2

Chemistry, 23.06.2019 13:00
Write the balanced chemical reaction for the formation of fe2(so4)3 from fe2o3 and so3 and determine how many moles of fe2(so4)3 are formed when 12.7 mol of so3 are reacted.
Answers: 1
You know the right answer?
What is the empirical formula of an unknown compound if was determined by a laboratory experiment th...
Questions

Biology, 01.07.2019 12:10

Geography, 01.07.2019 12:10



Biology, 01.07.2019 12:10

Geography, 01.07.2019 12:10

Geography, 01.07.2019 12:10

Geography, 01.07.2019 12:10


Physics, 01.07.2019 12:10


History, 01.07.2019 12:10

Chemistry, 01.07.2019 12:10