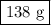Ca + 2h2o --> ca(oh)2 + h2
40g of ca is mixed with 100g of water. calculate mass of soluti...

Chemistry, 21.08.2019 20:30 jescanarias22
Ca + 2h2o --> ca(oh)2 + h2
40g of ca is mixed with 100g of water. calculate mass of solution left after reaction is complete.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 22:20
Calcium hydride (cah2) reacts with water to form hydrogen gas: cah2(s) + 2h2o(l) → ca(oh)2(aq) + 2h2(g) how many grams of cah2 are needed to generate 45.0 l of h2 gas at a pressure of 0.995 atm and a temperature of 32 °c?
Answers: 2

Chemistry, 22.06.2019 19:30
What is the mass of oxygen gas is consumed in a reaction that produces 4.60mol so2
Answers: 3

Chemistry, 22.06.2019 21:00
What is the chemical formula for the compound formed between sodium and flour one
Answers: 1

You know the right answer?
Questions


Computers and Technology, 12.08.2020 09:01

Biology, 12.08.2020 09:01




Mathematics, 12.08.2020 09:01

Chemistry, 12.08.2020 09:01

Mathematics, 12.08.2020 09:01

Biology, 12.08.2020 09:01


Mathematics, 12.08.2020 09:01




Biology, 12.08.2020 09:01