
Chemistry, 25.08.2019 21:10 Andrebutrus
***30
if 25.0 ml of a 0.100 m aqueous sodium hydroxide is mixed with 25.0 ml of a 0.100 m aqueous hydrochloric acid in a calorimeter at an initial temperature of 23.0 degrees celsius, what is the enthalpy change of this reaction if the final temperature reached in the calorimeter is 25.5 degrees celsius?
naoh + hcl yields nacl + h2o

Answers: 3
Another question on Chemistry



Chemistry, 22.06.2019 14:00
The content of manganese (mn) in steel was determined spectrophotometrically and with the use of the standard addition method. an unknown sample of mn from a digested steel sample gave an absorbance of 0.185 when analyzed spectrophotometrically. when 5.00 ml of solution containing 95.5 ppm mn was added to 50.0 ml of the unknown steel solution (digested sample), the absorbance was 0.248. calculate the concentration, in parts-per-million (ppm), of mn in the digested steel sample solution.
Answers: 3

Chemistry, 22.06.2019 17:10
Increasing the substrate concentration in an enzymatic reaction could overcome which of the following? a) the need for a coenzymeb) allosteric inhibitionc) competitive inhibitiond) insufficient cofactors
Answers: 1
You know the right answer?
***30
if 25.0 ml of a 0.100 m aqueous sodium hydroxide is mixed with 25.0 ml of a 0.100 m aqu...
if 25.0 ml of a 0.100 m aqueous sodium hydroxide is mixed with 25.0 ml of a 0.100 m aqu...
Questions

Mathematics, 20.05.2020 17:58

Mathematics, 20.05.2020 17:58


Mathematics, 20.05.2020 17:58

Physics, 20.05.2020 17:58


Mathematics, 20.05.2020 17:58

Mathematics, 20.05.2020 17:58


Mathematics, 20.05.2020 17:58

Health, 20.05.2020 17:58


Physics, 20.05.2020 17:58


History, 20.05.2020 17:58

Mathematics, 20.05.2020 17:58

Mathematics, 20.05.2020 17:58


Geography, 20.05.2020 17:58

History, 20.05.2020 17:58

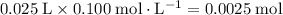 of both NaOH and HCl are available. As a result, 0.0025 moles of the reaction would have taken place.
of both NaOH and HCl are available. As a result, 0.0025 moles of the reaction would have taken place.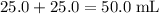 .
.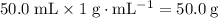 .
.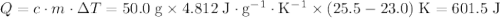
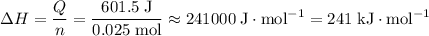 .
.

