
Study figure 13 and answer these questions. (figure is shown at the bottom)
each hydrogen atom ?
each oxygen atom ?
(s) is (are) needed to complete hydrogen's outer shell?
pair(s) is (are) being shared between the hydrogen atoms and the bonded oxygen?
(s) exist(s) between the hydrogen and the oxygen atom?
there electrons are in oxygen's outer orbit?
the inner/outer orbit of oxygen has two electrons?

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 10:10
For the reaction, 4 a(g) + 3 b(g) => 2 c(g), the following data were obtained at constant temperature. experiment initial[a],mol/l initial [b],mol/l initial rate,m/min 1 0.200 0.150 5.00 2 0.400 0.150 10.0 3 0.200 0.300 10.0 4 0.400 0.300 20.0 which of the following is the correct rate law for the reaction? 1. rate = k[a]2[b]2 2. rate = k[a][b] 3. rate = k[a]2[b] 4. rate = k[a][b]2
Answers: 3


Chemistry, 22.06.2019 17:30
A650 ml sodium bromine solution has a bromide ion concentration of 0.245 m. what is the mass (g) of sodium bromide in solution? a) 103.b)0.00155.c)16400.d) 16.4.e) 0.159
Answers: 2
You know the right answer?
Study figure 13 and answer these questions. (figure is shown at the bottom)
each hydrogen atom...
each hydrogen atom...
Questions

Mathematics, 13.02.2021 22:10

Mathematics, 13.02.2021 22:10

Social Studies, 13.02.2021 22:10

English, 13.02.2021 22:10






Social Studies, 13.02.2021 22:10

Mathematics, 13.02.2021 22:10


Mathematics, 13.02.2021 22:10

History, 13.02.2021 22:10


Mathematics, 13.02.2021 22:10

Mathematics, 13.02.2021 22:10

Mathematics, 13.02.2021 22:10


Mathematics, 13.02.2021 22:10

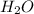 ) molecule. As atomic number of hydrogen is 1 so, its electronic distribution is 1 only. Also, atomic mass of a hydrogen atom is 1.
) molecule. As atomic number of hydrogen is 1 so, its electronic distribution is 1 only. Also, atomic mass of a hydrogen atom is 1. 

