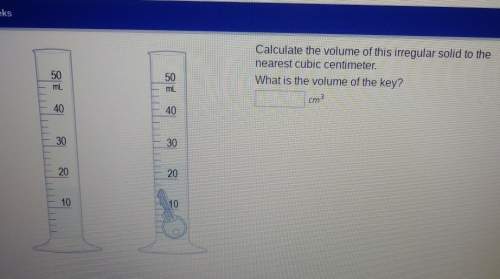
Calculate the volume of this irregular solid to the nearest cubic centimeter
...


Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 06:30
The following reaction shows the products when sulfuric acid and aluminum hydroxide react. al(oh)3 + h2so4 → al2(so4)3 + h2o the table shows the calculated amounts of reactants and products when the reaction was conducted in a laboratory. sulfuric acid aluminum hydroxide initial amount of reactant 40 g 15 g theoretical yield of water from reactant 14.69 g 10.38 g what is the approximate amount of the leftover reactant? 11.73 g of sulfuric acid 10.33 g of sulfuric acid 11.12 g of aluminum hydroxide 13.67 g of aluminum hydroxide
Answers: 3

Chemistry, 22.06.2019 10:00
Americium-241 undergoes fission to produce three neutrons per fission event. if a neutron-absorbing material is mixed in with this sample so that the rate of neutron production drops down to 1.8 neutrons per fission event, which will be effective at achieving a critical mass? check all that apply. remove a deflective shield surrounding the sample. remove absorbent material mixed in with the sample. compress the sample of americium-241.
Answers: 1

Chemistry, 22.06.2019 14:00
How does the presence of oxygen affect the chemical pathways used to extract energy from glucose?
Answers: 3
You know the right answer?
Questions

Mathematics, 31.08.2021 20:40


Biology, 31.08.2021 20:40

Mathematics, 31.08.2021 20:40



Mathematics, 31.08.2021 20:40



Biology, 31.08.2021 20:40

English, 31.08.2021 20:40


Physics, 31.08.2021 20:40

Mathematics, 31.08.2021 20:40

Mathematics, 31.08.2021 20:40


Business, 31.08.2021 20:40



Mathematics, 31.08.2021 20:40