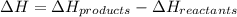Chemistry, 02.09.2019 20:10 herchellann302
When chlorine gas and hydrogen gas react to form hydrogen chloride, what will be the change of enthalpy of the reaction?

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 18:00
In a sample of oxygen gas at room temperature, the average kinetic energy of all the balls stays constant. which postulate of kinetic molecular theory best explains how this is possible? a. attractive forces between gas particles are negligible because the particles of an ideal gas are moving so quickly. b. collisions between gas particles are elastic; there is no net gain or loss of kinetic energy. c. gases consist of a large number of small particles, with a lot of space between the particles. d. gas particles are in constant, random motion, and higher kinetic energy means faster movement.
Answers: 1

Chemistry, 22.06.2019 05:50
Significant figures are digits read directly from the measuring instrument plus one more digit, which is __ by the observer.
Answers: 2

Chemistry, 22.06.2019 08:00
Define dew point. i am writing this part to be able to ask the question
Answers: 1

Chemistry, 22.06.2019 14:30
Is a pencil falling to the floor anon contact force, a force, or a contact force
Answers: 1
You know the right answer?
When chlorine gas and hydrogen gas react to form hydrogen chloride, what will be the change of entha...
Questions


History, 09.10.2019 15:20






Biology, 09.10.2019 15:20

Spanish, 09.10.2019 15:20

Mathematics, 09.10.2019 15:30



Mathematics, 09.10.2019 15:30


Mathematics, 09.10.2019 15:30

Physics, 09.10.2019 15:30


Computers and Technology, 09.10.2019 15:30


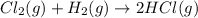
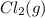 is 0 kJ/mol and
is 0 kJ/mol and 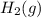 is also 0 kJ/mol.
is also 0 kJ/mol. 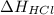 is -92.30 kJ/mol.
is -92.30 kJ/mol.