
Chemistry, 02.09.2019 21:20 reneewilliams20
The military has experimented with lasers that produce very intensive light when flourine combines explosively with hydrogen. what is the balanced equation for this reaction?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 15:20
An alloy contains 66 g of pure zinc. what is the percentage of zinc in the alloy? express your answer to two significant figures and include the appropriate units.
Answers: 1

Chemistry, 22.06.2019 20:00
Suppose that some of the compound spilled out of the crucible after it was heated. would that cause the percent by mass of water in the compound determined by the experiment to be too low, too high, or unchanged? briefly explain your answer.
Answers: 1

Chemistry, 22.06.2019 21:50
Answer the questions about this reaction: nai(aq) + cl2(g) → nacl(aq) + i2(g) write the oxidation and reduction half-reactions: oxidation half-reaction: reduction half-reaction: based on the table of relative strengths of oxidizing and reducing agents (b-18), would these reactants form these products? write the balanced equation: answer options: a. 0/na -> +1/na+1e- b. nai(aq) + cl2(g) → nacl(aq) + i2(g) c. +1/na+1e- -> 0 /na d. -1/2i -> 0/i2+2e- e. no f. 4nai(aq) + cl2(g) → 4nacl(aq) + i2(g) g. 2nai(aq) + cl2(g) → 2nacl(aq) + i2(g) h. 4nai(aq) + 2cl2(g) → 4nacl(aq) + 2i2(g) i. nai(aq) + cl2(g) → nacl(aq) + i2(g) j. 0/cl2+2e -> -1/2cl- k. yes
Answers: 1

Chemistry, 22.06.2019 22:40
Maleic acid is a carbon-hydrogen-oxygen compound used in dyeing and finishing fabrics and as a preservative of oils and fats.* in a combustion analysis, a 1.054-g sample of maleic acid yields 1.599 g of co_2 and 0.327 g of h2o.* when 0.609 g of maleic acid is dissolved in 25.00 g of glacial acetic acid, the freezing point drops by 0.82 c. maleic acid does not ionize in glacial acetic acid, which has a freezing-point depression constant of k_{\rm f}=3.90~^\circ {\rm c}/m.* in a titration experiment, a 0.4250- g sample of maleic acid is dissolved in water and requires 34.03 ml of 0.2152 m koh for its complete neutralization.* the ph of 0.215 g of maleic acid in 50.00 ml of aqueous solution is found to be 1.80.part adetermine the empirical formula for maleic acid. enter the atoms in order ofc, h, and o.part bdetermine the molecular formula for maleic acid. enter the atoms in order of c,h, and o.express your answer as a chemical formula.part cbased on the titration data, how many ionizable h atoms are in each molecule of maleic acid, c4h4o4? express your answer as an integer.i got the part a and b which is cho and c4h4o4 respectively, can anyone me for part c? the below is what i have so far from other qa'sfinding the empirical formula for maleic acid, needs this info"a combustion analysis a 1.054-g sample of maleic acid yields 1.599g of and 0.327g of ".the rest of your data is used to find molar mass, molecular formula, equivalent mass, # of h+ ions / molecule & katypically this should be: a 1.054-g sample of maleic acid yields 1.599g of co2 and 0.327g of h2osofind mass using molar masses1.599g of co2 @ (12.01 g carbon) / (44.01 g co2) = 0.436 grams of carbon0.327g of h2o @ (2.016 g hydrogen) / (18.01 g/mol h2o) = 0.037 grams of hydrogen1.054-g sample - 0.436 grams of carbon - 0.037 grams of hydrogen = 0.581 grams of oxygennow use molar masses to find moles0.436 grams of carbon @ 12.01 g/mol = 0.0363 mol c0.037 grams of hydrogen @ 1.008 g/mol = 0.0367 mol h0.581 grams of oxygen @ 16.00 g/mol = 0.0363 mol oyour empirical formula ischo and molecular is c4h4o4
Answers: 3
You know the right answer?
The military has experimented with lasers that produce very intensive light when flourine combines e...
Questions








Mathematics, 22.10.2020 20:01

History, 22.10.2020 20:01



Chemistry, 22.10.2020 20:01

Mathematics, 22.10.2020 20:01

Mathematics, 22.10.2020 20:01

Mathematics, 22.10.2020 20:01

Mathematics, 22.10.2020 20:01

Mathematics, 22.10.2020 20:01

Physics, 22.10.2020 20:01

Mathematics, 22.10.2020 20:01

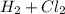 →
→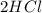
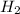 , chlorine gas is Cl_{2}, and the product of this reaction is hydrochloric acid, HCL.
, chlorine gas is Cl_{2}, and the product of this reaction is hydrochloric acid, HCL.


