

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 00:00
Which actions would increase the rate at salt dissolves in water? stir the water? crush the salt? use less water? heat the water? cool the salt
Answers: 3

Chemistry, 22.06.2019 04:00
Which of the following ocean acidification? are the most likely side effects of a ph less than 7.0 in the ocean b. more metal salts altering the ocean chemistry c. dissolution of the shells of marine organisms d. both a & b e. all of the above.
Answers: 3

Chemistry, 22.06.2019 06:30
The following reaction shows the products when sulfuric acid and aluminum hydroxide react. al(oh)3 + h2so4 → al2(so4)3 + h2o the table shows the calculated amounts of reactants and products when the reaction was conducted in a laboratory. sulfuric acid aluminum hydroxide initial amount of reactant 40 g 15 g theoretical yield of water from reactant 14.69 g 10.38 g what is the approximate amount of the leftover reactant? 11.73 g of sulfuric acid 10.33 g of sulfuric acid 11.12 g of aluminum hydroxide 13.67 g of aluminum hydroxide
Answers: 3

Chemistry, 22.06.2019 19:20
Consider hydrogen in an excited state n = 5n=5 that emits photons to reach the ground state. there are various possible transitions other than straight to the ground state that can occur; for example, it can drop to the n = 3n=3 state followed by the n = 3n=3 to the ground state transition. which of the possible transitions will result in the emission of a photon in the visible region?
Answers: 3
You know the right answer?
Calculate the ph of a buffer solution made from equal volumes of 0.3m hc2h3o2 and 0.6m nac2h3o2. (ka...
Questions





Biology, 02.11.2020 01:00




Mathematics, 02.11.2020 01:00

History, 02.11.2020 01:00


Business, 02.11.2020 01:00


Mathematics, 02.11.2020 01:00




Mathematics, 02.11.2020 01:00


Medicine, 02.11.2020 01:00

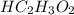 added =
added = 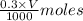
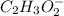 added =
added = 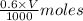
![pH=pK_{a}(HC_{2}H_{3}O_{2})+log[\frac{moles of C_{2}H_{3}O_{2}^{-}}{moles of HC_{2}H_{3}O_{2}}]](/tpl/images/0223/1512/efb5e.png)
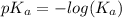
![pH=-log(1.8\times 10^{-5})+log[\frac{\frac{0.6\times V}{1000}}{\frac{0.3\times V}{1000}}]](/tpl/images/0223/1512/88cba.png)
![pH=-log(1.8\times 10^{-5})+log[\frac{0.6}{0.3}]](/tpl/images/0223/1512/f37b2.png)


