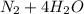Chemistry, 05.09.2019 20:30 hamadehassan
Write half-reactions for the oxidation and reduction process for each of the following.
a. fe2+ + mno4 - fe3+ + mn2+
b. sn2+ + io3 - sn4+ + i-
c. s2- + no3 - s + no
d. nh3 + no2 n2 + h2o

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 04:50
Compare the equilibrium constants for the systems shown in the table. which favors products the most? which favors products the least? rank these systems in order from most to least in terms of favoring products rather than reactants. d > b > a > c c > a > b > d b > c > d > a a > d > c > b
Answers: 1

Chemistry, 22.06.2019 06:00
24. a sports ball is inflated to an internal pressure of 1.85 atm at room temperature (25 °c). if the ball is then played with outside where the temperature is 7.5 °c, what will be the new pressure of the ball? assume the ball does not change in volume nor does any air leak from the ball a) 0.555 atm b) 1.74 atm c) 1.85 atm d) 1.97 atm
Answers: 2


You know the right answer?
Write half-reactions for the oxidation and reduction process for each of the following.
a. fe...
a. fe...
Questions

Mathematics, 23.05.2020 01:01


Mathematics, 23.05.2020 01:01


Mathematics, 23.05.2020 01:01

Chemistry, 23.05.2020 01:01

History, 23.05.2020 01:01

Mathematics, 23.05.2020 01:01

Chemistry, 23.05.2020 01:01



Social Studies, 23.05.2020 01:01

History, 23.05.2020 01:01


History, 23.05.2020 01:01





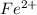 ⇒
⇒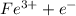
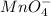 + 2
+ 2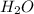 + 3
+ 3 ⇒
⇒ 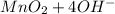
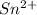 ⇒
⇒ 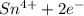
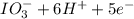 ⇒
⇒ 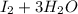
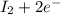 ⇒
⇒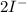
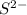 ⇒
⇒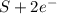
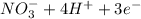 ⇒
⇒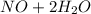
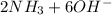 ⇒
⇒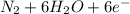
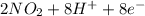 ⇒
⇒