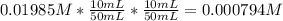Chemistry, 06.09.2019 18:30 angelteddy033
The molar concentration (m) of a solution prepared by dissolving 0.2362g of cr(no3)3 in a 50-ml volumetric flask is 0.01985m, where the molecular weight for cr(no3)3 = 238.01g/mol.
a. suppose you want to prepare another solution containing chromium nitrate that is 25 times less concentrated than the one prepared above. given a choice of 10-ml and 5-ml pipets and 50-ml and 100-ml volumetric flasks, explain how you would proceed in preparing the new diluted solution. in addition, calculate the concentration for the new diluted solution. show all work. your final value should have the correct unit and number of significant figures. hint: you will most likely need two dilution steps in order to obtain the desired concentration. note: you may not reuse the same pipet or combine different pipets within the same dilution step. you may reuse the pipet and/or volumetric flask in the different dilution step.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 07:30
Using data from seismic waves, geologists have learned that earth’s interior is made up of several
Answers: 1


Chemistry, 22.06.2019 14:30
The valence of aluminum is +3, and the valence of the chlorine is -1. the formula fir the aluminum chloride is correctly written as
Answers: 2

Chemistry, 22.06.2019 15:00
Answer explain why it is not possible to deduce a complete order of reactivity.
Answers: 3
You know the right answer?
The molar concentration (m) of a solution prepared by dissolving 0.2362g of cr(no3)3 in a 50-ml volu...
Questions

Mathematics, 31.10.2019 09:31

Health, 31.10.2019 09:31



Biology, 31.10.2019 09:31


Mathematics, 31.10.2019 09:31


Biology, 31.10.2019 09:31

Health, 31.10.2019 09:31

Social Studies, 31.10.2019 09:31



Chemistry, 31.10.2019 09:31


Mathematics, 31.10.2019 09:31