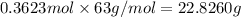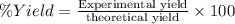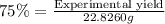Chemistry, 09.09.2019 17:20 victoria1831
If the percent yield for the following reaction is 75.0%, and 25.0 g of no₂ are consumed in the reaction, how many grams of nitric acid, hno₃(aq) are produced?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 09:00
George is a dalmatian puppy. describe what happens to light that allows you to see george’s black and white coat.
Answers: 1

Chemistry, 22.06.2019 17:30
Upon decomposition, one sample of magnesium fluoride produced 1.65 kg of magnesium and 2.56 kg of fluorine. a second sample produced 1.32 kg of magnesium. part a how much fluorine (in grams) did the second sample produce?
Answers: 2

Chemistry, 22.06.2019 19:10
Astudent completes a titration by adding 12.0 milliliters of naoh(aq) of unknown concentration to 16.0 milliliters of 0.15 m hcl(aq). what is the molar concentration of the naoh(aq)? 1)5.0 m 2)0.20 m 3)0.11 m 4)1.1 m
Answers: 1

Chemistry, 23.06.2019 00:00
Before it was launched, a helium-filled balloon had a pressure of 201 kpa at a temperature of 27°c. at an altitude of 15,000 m, the pressure had decreased to 2.5 kpa and the temperature had dropped to -14 °c. the volume of the balloon increased to 59.3 m3. what is the original volume of the balloon? 13 m3 0.85 m3 0.077 m3 1.17 m3
Answers: 3
You know the right answer?
If the percent yield for the following reaction is 75.0%, and 25.0 g of no₂ are consumed in the reac...
Questions


Chemistry, 07.11.2020 07:50

Mathematics, 07.11.2020 07:50

Mathematics, 07.11.2020 07:50


Mathematics, 07.11.2020 07:50

English, 07.11.2020 07:50

Mathematics, 07.11.2020 07:50

Arts, 07.11.2020 07:50


Social Studies, 07.11.2020 07:50

English, 07.11.2020 07:50


Mathematics, 07.11.2020 07:50




Mathematics, 07.11.2020 07:50

Health, 07.11.2020 07:50

Social Studies, 07.11.2020 07:50

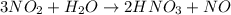
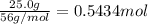
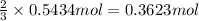 of nitric acid.
of nitric acid.