
Chemistry, 09.09.2019 23:30 jennypenny123
Which of the following statements are true concerning ionic bonding? select one: a. ionic bonding occurs between a metal, which has a high affinity for electrons, and a nonmetal, which loses electrons relatively easy. b. cacl2 forms because ca2+ is always a more stable species than the calcium atom alone. c. compounds with ionic bonds tend to have low melting points. d. the electronegativity difference between the bonding atoms of ionic compounds is small since the electrons are not shared but rather held together by electrostatic forces. e. all of the above statements are false.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 21:00
Read these sentences from the text. near the equator, the tropics receive the most rain on a consistent basis. as a result, the fresh water falling into the ocean decrease the salinity of the surface water in that region. [. .] . . as the salt content of sea water increases, so does its density. what can you infer about how rain affects the density of surface water near the equator?
Answers: 1

Chemistry, 22.06.2019 10:00
Miner's coal distributors does not mine coal itself, nor does it even store or handle the coal. instead, miner's solicits orders for low sulfur coal from other firms, then purchases the required amount from suppliers and directs them to ship the coal to its customers. what is miner's
Answers: 1

Chemistry, 22.06.2019 10:00
Suppose the universe were completely empty except for one object-a solid sphere moving through space of 100 km/s. what sort of path would the object be moving in? explain your answer
Answers: 1

Chemistry, 22.06.2019 11:00
The diagram below shows the different phase transitions that occur in matter. which arrow represents the transition in which dew is formed?
Answers: 1
You know the right answer?
Which of the following statements are true concerning ionic bonding? select one: a. ionic bonding...
Questions

















Biology, 12.11.2019 23:31



 forms because
forms because 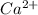 is always a more stable species than the calcium atom alone
is always a more stable species than the calcium atom alone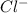 is an anion with oxidation state of -1. Thus they combine and their oxidation states are exchanged and written in simplest whole number ratios to give neutral
is an anion with oxidation state of -1. Thus they combine and their oxidation states are exchanged and written in simplest whole number ratios to give neutral ![[Ca]=1s^22s^22p^63s^23p^64s^2](/tpl/images/0226/1839/cdc07.png)
![[Ca^{2+}]=1s^22s^22p^63s^23p^6](/tpl/images/0226/1839/04411.png)
![[Cl]=1s^22s^22p^63s^23p^5](/tpl/images/0226/1839/7a3e4.png)
![[Cl^-]=1s^22s^22p^63s^23p^6](/tpl/images/0226/1839/bea8e.png)



