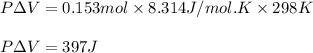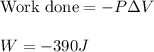Chemistry, 13.09.2019 20:30 willcohen42
Apellet of zn of mass 10.0g is dropped into a flaskcontaining
dilute h2so4 at a pressure of p=1.00 bar and
temperature of 298k. what is the reaction thatoccures? calculate w
for the process.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 22:50
2. you__turn left on a red light if you are in the left-most lane of a one-way street, you're turning into the left-most lane of a one-way street, and no nearby sign prohibits the turn.
Answers: 2

Chemistry, 22.06.2019 05:10
How many miles of water are produced if 5.43 mol pbo2 are consumed
Answers: 1

Chemistry, 22.06.2019 07:40
The formation of a solid, also known as a is an indication of a chemical change. precipitate particulate particle powder
Answers: 3

Chemistry, 22.06.2019 14:30
Consider the reduction reactions and their equilibrium constants. cu+(aq)+e−↽−−⇀cu(s)pb2+(aq)+2e−↽−−⇀pb(s)fe3+(aq)+3e−↽−−⇀fe(=6.2×108=4.0×10−5=9.3×10−3 cu + ( aq ) + e − ↽ − − ⇀ cu ( s ) k =6.2× 10 8 pb 2 + ( aq ) +2 e − ↽ − − ⇀ pb ( s ) k =4.0× 10 − 5 fe 3 + ( aq ) +3 e − ↽ − − ⇀ fe ( s ) k =9.3× 10 − 3 arrange these ions from strongest to weakest oxidizing agent.
Answers: 3
You know the right answer?
Apellet of zn of mass 10.0g is dropped into a flaskcontaining
dilute h2so4 at a pressure of p=...
dilute h2so4 at a pressure of p=...
Questions




English, 14.09.2021 19:00

Chemistry, 14.09.2021 19:00



Chemistry, 14.09.2021 19:00


World Languages, 14.09.2021 19:00






Mathematics, 14.09.2021 19:00


Physics, 14.09.2021 19:00


Mathematics, 14.09.2021 19:00

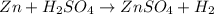
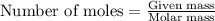
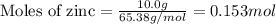
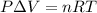
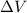 = Change in volume of the gas
= Change in volume of the gas