
Chemistry, 14.09.2019 00:30 goldenhawk239
The vapour pressure of water at 20 c is 2.34 kpa. given that the heat of vaporisation is 2537.4 kj/kg, use the clausius-clapeyron equation to give the vapour pressure at 40 c.

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 06:30
Predict whether the changes in enthalpy, entropy, and free energy will be positive or negative for the boiling of water, and explain your predictions. how does temperature affect the spontaneity of this process?
Answers: 1


Chemistry, 22.06.2019 23:00
What is a substance? a. a physical property of matter b. a chemical property of matter c. an element or compound that cannot be physically separated d. characteristics used to tell the difference between mixtures
Answers: 1
You know the right answer?
The vapour pressure of water at 20 c is 2.34 kpa. given that the heat of vaporisation is 2537.4 kj/k...
Questions

Mathematics, 07.06.2021 01:00

Biology, 07.06.2021 01:00

Chemistry, 07.06.2021 01:00


Mathematics, 07.06.2021 01:00




Mathematics, 07.06.2021 01:00

Mathematics, 07.06.2021 01:00

Social Studies, 07.06.2021 01:00







History, 07.06.2021 01:00


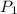 = 2.34 kPa = 2.34 \times 1000 Pa = 2340 Pa = 0.0231 atm
= 2.34 kPa = 2.34 \times 1000 Pa = 2340 Pa = 0.0231 atm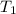 = (20 + 273) K = 293 K
= (20 + 273) K = 293 K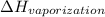 = 2537.4 kJ/kg = 2537400 J/kg
= 2537.4 kJ/kg = 2537400 J/kg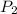 = ?,
= ?, 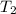 = (40 + 273) K = 313 K
= (40 + 273) K = 313 K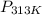 =
= ![P_{293} Exp[\frac{-\Delta H_{v}}{R} (\frac{1}{313} - \frac{1}{293})]](/tpl/images/0230/4993/fdff5.png)
![0.0213 Exp [\frac{-2537400 J/kg}{8.314 J/mol} (\frac{1}{313} - \frac{1}{293})K]](/tpl/images/0230/4993/b4000.png)
 atm
atm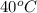 is 18846.45 kPa.
is 18846.45 kPa.

