
Chemistry, 14.09.2019 02:20 winstonbendariovvygn
You have a sample of water that contains the organic compound
c7h12on2 at a concentration of 50
mg/l. the compound can be oxidized by bacteria to form carbon
dioxide, water, and ammonia. how many mg/l of oxygen is needed to
biodegrade the compound? note: determine only carbonaceous
demand.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 22:30
1.aluminum chloride (alcl3), and sodium hydroxide (naoh) can react to form aluminum hydroxide (al(oh)3) and sodium chloride (nacl). you have 13.4 g of aluminum chloride and 10.0 g of sodium hydroxide. answer the following questions: •what is the balanced equation for this reaction? •if you use all 13.4 g of aluminum chloride, how many grams of aluminum hydroxide can be formed? work must be shown to earn credit •if you use all 10.0 g of sodium hydroxide, how many grams of aluminum hydroxide can be formed? work must be shown to earn credit •how many grams of aluminum hydroxide will actually be made? which reagent is limiting? explain your answer.
Answers: 1

Chemistry, 22.06.2019 09:00
George is a dalmatian puppy. describe what happens to light that allows you to see george’s black and white coat.
Answers: 1

Chemistry, 22.06.2019 12:20
Consider the reaction of a(g) + b(g) + c(g) => d(g) for which the following data were obtained: experiment initial [a], mol/l initial [b], mol/l initial [c], mol/l initial rate, mol/l.s 1 0.0500 0.0500 0.0100 6.25 x 10^-3 2 0.100 0.0500 0.0100 2.50 x 10^-2 3 0.100 0.100 0.0100 1.00 x 10^-1 4 0.0500 0.0500 0.0200 6.25 x 10^-3 what is the rate law for the reaction?
Answers: 3

You know the right answer?
You have a sample of water that contains the organic compound
c7h12on2 at a concentration of 5...
c7h12on2 at a concentration of 5...
Questions

Social Studies, 21.04.2020 18:30

History, 21.04.2020 18:30










Mathematics, 21.04.2020 18:30


English, 21.04.2020 18:30

Biology, 21.04.2020 18:30

Mathematics, 21.04.2020 18:30


Mathematics, 21.04.2020 18:30

Biology, 21.04.2020 18:30

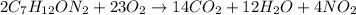
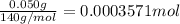
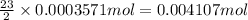 oxygen gas.
oxygen gas.

