Chemistry, 14.09.2019 09:30 litwork5885
Hexane is a common laboratory solvent and a component of petrol. its combustion reaction is as follows: 2 c6h14 (9) + 19 02(9) +12 co2(g) + 14 h20(1) arh=-8326 kj mol-1 use the reaction enthalpy value to calculate the heat in (kj) released by combustion of 0.121 mol of hexane. (give your answer as a positive number to at least three significant figures and do not include the units in the answer box)

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 05:30
The table describes how some substances were formed substance 19 description formed by boiling pure water formed by combining three hydrogen atoms to every nitrogen atom formed by adding 5 g of sugar to 1 l of water formed by compressing carbon under high pressure based on the given descriptions, which substance is most likely a mixture?
Answers: 1

Chemistry, 22.06.2019 07:30
The volume of helium in a blimp is 6.28 x 10^9 millimeters. the density of helium in the blimp is .1786 kilogram/meter^3. find the mass of the helium in the blimp.
Answers: 1


Chemistry, 22.06.2019 22:30
Gusing the milligrams of ascorbic acid you entered above, the ratio of total sample volume to aliquot volume, and the total milligrams of the vitamin c tablet that you dissolved, calculate the mass of ascorbic acid in the vitamin c tablet for each trial. do this by scaling up to find the amount (mg) of ascorbic acid in your 250 ml flask. enter your calculated mass of ascorbic acid in the vitamin c tablet, for each trial. be sure to enter your calculated mass in the corresponding order that you entered your milligrams of ascorbic acid. the milligrams of ascorbic acid you entered for entry #1 previously should correspond to the mass of ascorbic acid that you enter for entry #1 here.
Answers: 1
You know the right answer?
Hexane is a common laboratory solvent and a component of petrol. its combustion reaction is as follo...
Questions



French, 21.05.2020 03:11

History, 21.05.2020 03:11


Mathematics, 21.05.2020 03:11



Mathematics, 21.05.2020 03:11

Mathematics, 21.05.2020 03:11

Computers and Technology, 21.05.2020 03:11



History, 21.05.2020 03:11


Biology, 21.05.2020 03:11

Advanced Placement (AP), 21.05.2020 03:11

History, 21.05.2020 03:11


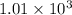
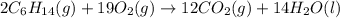
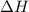 = -8326 kJ/mol
= -8326 kJ/mol