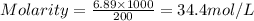Calculating molarity using solute moles a chemist prepares a solution of sodium chloride (nacl) by measuring out 6.89 mol of sodium chloride into a 200 ml volumetric flask and filling the the mark with water. calculate the concentration in mol/l of the chemist's sodium chloride solution. round your answer to 3 significant digits. x 5 ? explanation check

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 09:20
What happened to the amount of carbon dioxide in the atmosphere from 2010–2017?
Answers: 1

Chemistry, 22.06.2019 12:20
Achemistry student weighs out 0.306 g of citric acid (h3c6h5o7), a triprotic acid, into a 250 ml volumetric flask and dilutes to the mark with distilled water. he plans to titrate the acid with 0.1000 m naoh solution. calculate the volume of naoh solution the student will need to add to reach the final equivalence point. be sure your answer has the correct number of significant digits.
Answers: 3

Chemistry, 22.06.2019 13:00
One of the hopes for solving the world's energy problem is to make use of the fusion reaction 21h +31h --> 42he + 10n + energy how much energy is released when 1 mol of deuterium is fused with 1 mol of tritium according to the above reaction? the masses of the atoms and the neutrons are as follows: 21h = 2.0140 amu 31h = 3.01605 amu 42he = 4.002603 amu 10n = 1.008665 amu. the speed of light is 2.9979 x 108 m/s.
Answers: 1
You know the right answer?
Calculating molarity using solute moles a chemist prepares a solution of sodium chloride (nacl) by m...
Questions




Mathematics, 23.07.2019 16:30


Mathematics, 23.07.2019 16:30

History, 23.07.2019 16:30




History, 23.07.2019 16:30



Mathematics, 23.07.2019 16:30

History, 23.07.2019 16:30

Social Studies, 23.07.2019 16:30

Biology, 23.07.2019 16:30

History, 23.07.2019 16:30

Mathematics, 23.07.2019 16:30

Social Studies, 23.07.2019 16:30

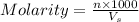
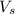 = volume of solution in ml
= volume of solution in ml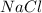 = 6.89
= 6.89