
Chemistry, 16.09.2019 18:00 coontcakes
Octane is a hydrocarbon that is found in gasoline. complete combustion of octane produces 8 l of carbon dioxide for every 9 l of water vapor (both measured at the same temperature and pressure). what is the ratio of carbon atoms to hydrogen atoms in a molecule of octane?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 03:30
What is the relationship of air masses and the temperature of oceans?
Answers: 1

Chemistry, 22.06.2019 20:40
What effect would average population growth have on land usage? a. urban use of land would rise to more than 30 percent of available land. b. industrial use of land would rise to more than 30 percent of available land. c. the percentage of available land used as cropland would stay the same. d. cropland would fall to about 10 percent of available land.
Answers: 1

Chemistry, 23.06.2019 06:10
2. what two items do autotrophs take from the environment to produce their food? 3. what are the two items that are released during transpiration from leaves? 4. what are the two membranes of the system? a.what are the two stages of photosynthesis? what are the two parts of photosynthesis?
Answers: 2

Chemistry, 23.06.2019 14:00
In 1901, thomas edison invented the nickel-iron battery. the following reaction takes place in the battery. fe(s) + 2 nio(oh)(s) + 2 h2o(l) fe(oh)2(s) + 2 ni(oh)2(aq) how many mole of fe(oh)2, is produced when 6.00 mol fe and 8.45 mol nio(oh) react?
Answers: 1
You know the right answer?
Octane is a hydrocarbon that is found in gasoline. complete combustion of octane produces 8 l of car...
Questions



Arts, 29.10.2021 14:00


Business, 29.10.2021 14:00



Computers and Technology, 29.10.2021 14:00


English, 29.10.2021 14:00

History, 29.10.2021 14:00

Mathematics, 29.10.2021 14:00


Biology, 29.10.2021 14:00

Computers and Technology, 29.10.2021 14:00

Mathematics, 29.10.2021 14:00

Mathematics, 29.10.2021 14:00



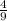
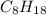 ).
).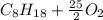 ⇒
⇒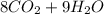
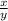 , where x and y comes from
, where x and y comes from 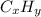 , or
, or 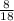 , which simplified is
, which simplified is 

