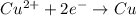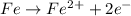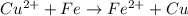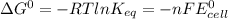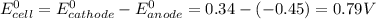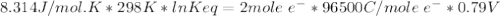Chemistry, 19.09.2019 04:20 IkarosSakurai
Agalvanic cell consists of a iron electrode in 1 m fe(no3)2 and a copper electrode in 1 m cu(no3)2. what is the equilibrium constant for this reaction at 25oc?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 02:30
When you perform this reaction, what could remain at the end of the reaction? check all that apply. excess reactant aqueous copper chloride excess reactant aluminum oxygen product solid copper carbon dioxide product aqueous aluminum chloride water
Answers: 2

Chemistry, 22.06.2019 05:00
As you watch a surfer ride a wave towards the shoreline, what is the shoreline? a) displacement reference b) reference point c) coordinate plane d) cartesian boundary
Answers: 1

Chemistry, 22.06.2019 07:10
Provide a stepwise curved arrow mechanism that fully explains the outcome of the reaction shown below. oh нао* heat он
Answers: 2

Chemistry, 22.06.2019 10:40
Ammonia and oxygen react to form nitrogen monoxide and water, like this: 4nh3 (g) + 5o2 (g) → 4no (g) + 6h2o (g) also, a chemist finds that at a certain temperature the equilibrium mixture of ammonia, oxygen, nitrogen monoxide, and water has the following composition: compound pressure at equilibrium nh3 65.1atm o2 31.3atm no 62.7atm h2o 65.8atm compound pressure at equilibrium nh3 65.3 atm o2 7.79 atm no 12.1 atm h2o 65.8 atm calculate the value of the equilibrium constant kp for this reaction. round your answer to 2 significant
Answers: 2
You know the right answer?
Agalvanic cell consists of a iron electrode in 1 m fe(no3)2 and a copper electrode in 1 m cu(no3)2....
Questions


Mathematics, 07.10.2021 16:30

History, 07.10.2021 16:30

Mathematics, 07.10.2021 16:30

Business, 07.10.2021 16:30

History, 07.10.2021 16:30

Biology, 07.10.2021 16:30


English, 07.10.2021 16:30

Physics, 07.10.2021 16:30


SAT, 07.10.2021 16:30