Chemistry, 23.09.2019 21:10 sparky1234
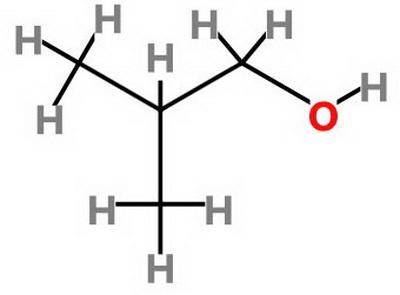
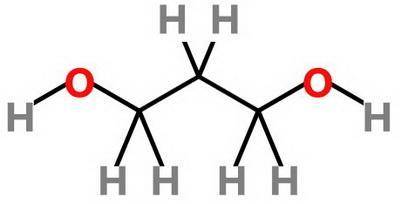
Which statement about 2‑methyl‑1‑propanol, (ch3)2chch2oh , and 1,3‑propanediol, hoch2ch2ch2oh is true? 2‑methyl‑1‑propanol is more soluble in water than 1,3‑propanediol because 2‑methyl‑1‑propanol has a smaller molecular mass. 2‑methyl‑1‑propanol is more soluble in water than 1,3‑propanediol because 2‑methyl‑1‑propanol forms fewer hydrogen bonds with water. 1,3‑propanediol is more soluble in water than 2‑methyl‑1‑propanol because 1,3‑propanediol has a smaller molecular mass. 1,3‑propanediol is more soluble in water than 2‑methyl‑1‑propanol because 1,3‑propanediol can form multiple hydrogen bonds with water.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 01:30
Sulfuric acid (a component of acid rain) reacts with limestone (calcium carbonate) to produce calcium sulfate and carbon dioxide. this damages buildings and statues made of limestone. which solution of sulfuric acid will damage these structures more quickly? a. 0.001% b. 0.005% c. 0.010% d. 0.015%
Answers: 3


Chemistry, 22.06.2019 21:30
In science class richard learns that a substance has a boiling point of 230 fahrenheit his teacher ask him to convert this temperature to degrees celsius what is the boiling point of his substance in degrees celsius
Answers: 3

Chemistry, 22.06.2019 23:30
Imagine a small synthetic vesicle made from pure phospholipids enclosing an interior lumen containing 1 mm glucose and 1 mm sodium chloride. if the vesicle is placed in pure water, which of the following happens faster? a. na+ diffuses out. b. cl– diffuses out. c. h2o diffuses in. d. glucose diffuses out. e. sodium chloride diffuses out.
Answers: 3
You know the right answer?
Which statement about 2‑methyl‑1‑propanol, (ch3)2chch2oh , and 1,3‑propanediol, hoch2ch2ch2oh is tru...
Questions


English, 20.01.2020 07:31

Mathematics, 20.01.2020 07:31


Chemistry, 20.01.2020 07:31


Chemistry, 20.01.2020 07:31

Mathematics, 20.01.2020 07:31

Chemistry, 20.01.2020 07:31

English, 20.01.2020 07:31

Physics, 20.01.2020 07:31


Mathematics, 20.01.2020 07:31

History, 20.01.2020 07:31


Physics, 20.01.2020 07:31

Mathematics, 20.01.2020 07:31


History, 20.01.2020 07:31