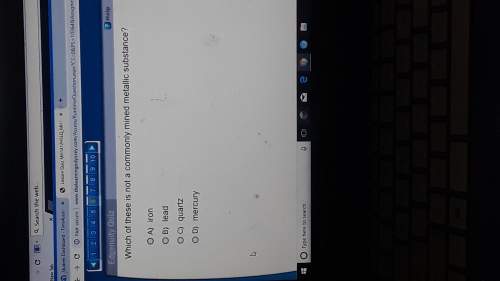
When methanol, ch3oh, is dissolve in water, a nonconducting solution reslults. when acetic acid, ch3cooh, diisolves in water, the solution is weakly conductng and acidic in nature. describe what happens upon dissolution in the two ases, and account for the different results

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 17:00
What is the empirical formula of vanadium 1 oxide given that 20.38 grams of vandium combines with oxygen to form 23.58 grams of the oxide
Answers: 1

Chemistry, 22.06.2019 00:00
1) these are barrel shaped microtubules in most animal cells, that organize the spindles during cell division
Answers: 1

Chemistry, 22.06.2019 09:00
This chart lists four kinds of polymers and their sources. what can be known about all four polymers, despite their differences? they come from living things. they share ionic carbon bonds. they are at least 100 monomers long. they are made of repeating subunits.
Answers: 2

Chemistry, 22.06.2019 11:30
Aperfume bottle is dropped in the corner of a room. the odor of the perfume can be detected on the other side of the room. which statement best describes this observation?
Answers: 2
You know the right answer?
When methanol, ch3oh, is dissolve in water, a nonconducting solution reslults. when acetic acid, ch3...
Questions

Mathematics, 02.06.2020 04:58









Mathematics, 02.06.2020 04:58


Arts, 02.06.2020 04:58


History, 02.06.2020 04:58


Social Studies, 02.06.2020 04:58