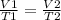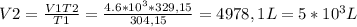Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 23:30
Calculate the expected ph values of the buffer systems from the experiments (a,b,c,d), using the henderson- hasselbalch equation, ph-pka+log[a-]/[ha]. use for pka values carbonic acid= 6.37, and acetic acid= 4.75.
Answers: 2

Chemistry, 22.06.2019 12:10
|using the periodic tablewarm-upuse the periodic table in the tools bar to answer the following questions.what elemental classification does oxygen belongto? done
Answers: 3


Chemistry, 22.06.2019 14:40
Choose an equation that represents an enzyme-catalyzed reaction. (a) enzyme + substrate → enzyme-substrate complex (b) enzyme + substrate ←−→ enzyme + products (c) enzyme + substrate ←−→ enzyme-substrate complex → enzyme + products (d) enzyme + substrate ←−→ enzyme-substrate complex → enzyme-substrate complex + products
Answers: 2
You know the right answer?
Aballoon filled with helium has a volume of 4.6 x 103 l at 31°c. what volume will the balloon occupy...
Questions

History, 03.12.2019 01:31



Mathematics, 03.12.2019 01:31

Mathematics, 03.12.2019 01:31

Spanish, 03.12.2019 01:31

Biology, 03.12.2019 01:31

Mathematics, 03.12.2019 01:31

History, 03.12.2019 01:31







Health, 03.12.2019 01:31




Advanced Placement (AP), 03.12.2019 01:31