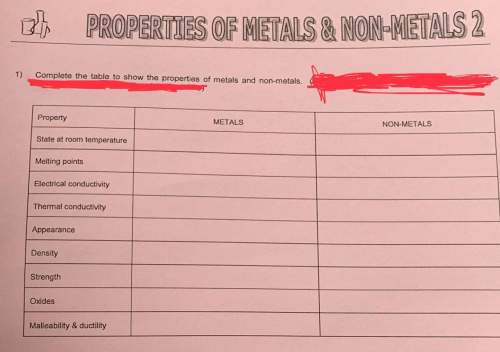
With everything and in advance i will mark branliest
...

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 22:30
1.aluminum chloride (alcl3), and sodium hydroxide (naoh) can react to form aluminum hydroxide (al(oh)3) and sodium chloride (nacl). you have 13.4 g of aluminum chloride and 10.0 g of sodium hydroxide. answer the following questions: •what is the balanced equation for this reaction? •if you use all 13.4 g of aluminum chloride, how many grams of aluminum hydroxide can be formed? work must be shown to earn credit •if you use all 10.0 g of sodium hydroxide, how many grams of aluminum hydroxide can be formed? work must be shown to earn credit •how many grams of aluminum hydroxide will actually be made? which reagent is limiting? explain your answer.
Answers: 1

Chemistry, 22.06.2019 00:50
If a reactant was removed, did the new equilibrium system shift to make more reactants or more products?
Answers: 1

Chemistry, 22.06.2019 08:30
What method(s) do plants use to obtain nitrogen? select all that apply. absorb it from the atmosphere use bacteria to convert nitrogen to usable form obtain usable nitrogen compounds from the soil absorb nitrogen from water taken in at the roots
Answers: 3

You know the right answer?
Questions

Mathematics, 28.01.2020 00:31



Mathematics, 28.01.2020 00:31


Mathematics, 28.01.2020 00:31

Mathematics, 28.01.2020 00:31


Mathematics, 28.01.2020 00:31

Mathematics, 28.01.2020 00:31

Physics, 28.01.2020 00:31

English, 28.01.2020 00:31

Mathematics, 28.01.2020 00:31

History, 28.01.2020 00:31



History, 28.01.2020 00:31

Mathematics, 28.01.2020 00:31