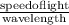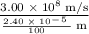Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 01:20
1. suppose a reaction mixture, when diluted with water, afforded 300 ml of an aqueous solution of 30 g of the reaction product malononitrile [ch2(cn)2], which is to be isolated by extraction with ether. the solubility of malononitrile in ether at room temperature is 20.0 g/100 ml, and in water is 13.3 g/100 ml. what weight of malononitrile would be recovered by extraction with (a) three 100-ml portions of ether and (b) one 300-ml portion of ether? suggestion: for each extraction, let x equal the weight extracted into the ether layer. in part (a), the concentration in the ether layer is x/100 and in the water layer is (30 x)/300; the ratio of these quantities is equal to k 20/13.3.
Answers: 2

Chemistry, 22.06.2019 21:00
Two nails have identical sizes and shapes. in one nail, 20 percent of the domains are lined up. in the other nail, 80 percent of the domains are lined up. which has stronger magnetic force? first answer gets brainliest!
Answers: 1

Chemistry, 22.06.2019 22:30
Molecular iodine, i2(g), dissociates into iodine atoms at 625 k with a first-order rate constant of 0.271 s−1. part a part complete what is the half-life for this reaction?
Answers: 3

Chemistry, 23.06.2019 10:30
Can anyone explain 1. review your spectrometry data and use the known elements to identify the star's composition. which unknown elements make up this star? justify your element selections. 2. in parts i and ii of the lab, what happened to the electrons of each element to produce the different colors of light? explain your answers using important terms from the lesson and information provided in the laboratory. 3. stars composed of heavier (more massive) elements are often slightly older than stars made predominantly from hydrogen and helium. based on your data, is the newly discovered star a younger star? explain your answer.
Answers: 2
You know the right answer?
What is the frequency of radiation whose wavelength is 2.40 x 10^-5 cm...
Questions

Physics, 01.12.2020 15:00

English, 01.12.2020 15:00

World Languages, 01.12.2020 15:00


SAT, 01.12.2020 15:00

English, 01.12.2020 15:00

English, 01.12.2020 15:10



Business, 01.12.2020 15:10

Chemistry, 01.12.2020 15:10

Business, 01.12.2020 15:10

Computers and Technology, 01.12.2020 15:10

English, 01.12.2020 15:10

English, 01.12.2020 15:10

Mathematics, 01.12.2020 15:10

English, 01.12.2020 15:10

Computers and Technology, 01.12.2020 15:10


English, 01.12.2020 15:10

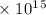 1\s with travelling in the light-medium.
1\s with travelling in the light-medium.