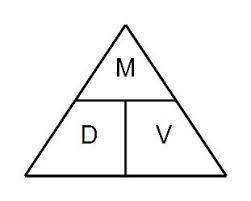
Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 07:00
Achemist wants to extract copper metal from copper chloride solution. the chemist places 0.50 grams of aluminum foil in a solution containing 0.75 grams of copper (ii) chloride. a single replacement reaction takes place. (ii) chloride. a single replacement reaction takes place. which statement explains the maximum amount of copper that the chemist can extract using this reaction? a) approximately 0.36 grams, because copper (ii) chloride acts as a limiting reactant b) approximately 1.8 grams, because copper (ii) chloride acts as a limiting reactant c) approximately 0.36 grams, because aluminum acts as a limiting reactant d) approximately 1.8 grams, because aluminum acts as a limiting reactant
Answers: 3

Chemistry, 22.06.2019 13:30
How many moles is 14.5 cm^3 of platinum? the density of platinum is 21.45 g/cm^3.
Answers: 1

Chemistry, 22.06.2019 23:30
If maltose undergoes hydrolysis what subunits does it results to?
Answers: 2

Chemistry, 23.06.2019 01:30
What happens to the concentration of hydronium ions as the ph of a solution increases? a. hydronium ion concentration stays the same b. hydronium ion concentration decreases c. hydronium ion concentration increases
Answers: 1
You know the right answer?
What is the volume of a 60.0 g object with a density of 2.12 g/cm3?...
Questions

Mathematics, 03.01.2020 17:31





Biology, 03.01.2020 17:31





Mathematics, 03.01.2020 17:31


Mathematics, 03.01.2020 17:31

Social Studies, 03.01.2020 17:31

Mathematics, 03.01.2020 17:31

History, 03.01.2020 17:31


Mathematics, 03.01.2020 17:31

Mathematics, 03.01.2020 17:31

Mathematics, 03.01.2020 17:31