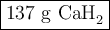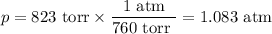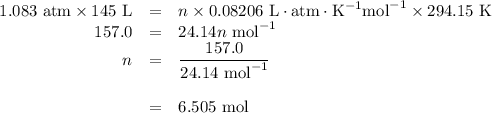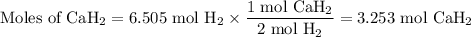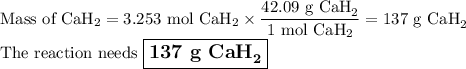Calcium hydride, cah2, reacts with water to form hydrogen gas:
cah2(s)+2h2o(l)→ca(oh)2(aq)+2...

Calcium hydride, cah2, reacts with water to form hydrogen gas:
cah2(s)+2h2o(l)→ca(oh)2(aq)+2h2(g)
this reaction is sometimes used to inflate life rafts, weather balloons, and the like, where a simple, compact means of generating h2 is desired.
how many grams of cah2 are needed to generate 145 l of h2 gas if the pressure of h2 is 823 torr at 21 ∘c?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 01:30
Reaction rate depends on how many molecules are coming into contact with each other with enough energy to react. increasing the temperature of the reactants will increase -
Answers: 3

Chemistry, 22.06.2019 05:40
Consider the elements bromine and chlorine; which elements has a larger ionic radius ?
Answers: 1

Chemistry, 22.06.2019 05:50
Calculate the number of molecules present in 0.750 mol of mgo.
Answers: 3

Chemistry, 22.06.2019 07:00
This image is an example of a(n) a) atom. b) compound. c) mixture. d) molecule.
Answers: 1
You know the right answer?
Questions

History, 17.01.2022 14:00

Chemistry, 17.01.2022 14:00


Mathematics, 17.01.2022 14:00

Mathematics, 17.01.2022 14:00

History, 17.01.2022 14:00


Mathematics, 17.01.2022 14:10

Mathematics, 17.01.2022 14:10

English, 17.01.2022 14:10


Biology, 17.01.2022 14:10


Mathematics, 17.01.2022 14:10



History, 17.01.2022 14:10