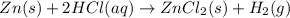When kay added 30 g of hydrochloric acid (hcl) to 65 g of zn, bubbles appeared and a white precipitate, zinc chloride (zncl2) was produced. she found that the mass of the zinc chloride was 93 g. since the law of conservation of matter stated that matter can't be created or destroyed, she was puzzled about the difference in mass. what is the best explanation for the missing 2 g?
a) 2 g of zinc disappeared.
b) 2 g of hydrogen gas were produced.
c) kay didn't correctly mass the precipitate.
d) kay didn't collect all of the precipitate.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 15:30
Anurse practitioner prepares an injection of promethazine, an antihistamine used to treat allergic rhinitis. if the stock bottle is labeled 25 mg/ml and the order is a dose of 11.0 mg , how many milliliters will the nurse draw up in the syringe?
Answers: 3

Chemistry, 22.06.2019 07:00
At 450 mm hg a gas has a volume of 760 l, what is its volume at standard pressure
Answers: 2

Chemistry, 22.06.2019 16:50
Answer asap need it by wednesday morning calculate the ph of 0.02m hcl best answer will be brainliest
Answers: 1

You know the right answer?
When kay added 30 g of hydrochloric acid (hcl) to 65 g of zn, bubbles appeared and a white precipita...
Questions


Business, 09.06.2021 01:00

Mathematics, 09.06.2021 01:00


Mathematics, 09.06.2021 01:00

Chemistry, 09.06.2021 01:00

Mathematics, 09.06.2021 01:00




Mathematics, 09.06.2021 01:00

Social Studies, 09.06.2021 01:00



Mathematics, 09.06.2021 01:00

English, 09.06.2021 01:00


Chemistry, 09.06.2021 01:00