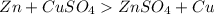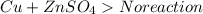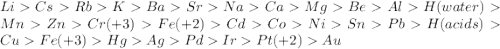Chemistry, 05.10.2019 13:30 dragpack70
Which of the following will be more reactive than magnesium ()

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 22:00
Fission of uranium-235 products energy and a. isotopes of smaller elements b. isotopes of larger elements c. lighter isotopes of uranium d. heavier isotopes of uranium
Answers: 3

Chemistry, 22.06.2019 03:30
Melting and boiling are endothermic processes. this means that these processes absorb energy from their surroundings in order to occur. use this information and the data you collected in the phase change gizmo to describe what happens to the temperature of water when you boil it, then explain why this result occurs.
Answers: 2

Chemistry, 22.06.2019 22:30
Vi limitens. vastery test select the correct answer. which statement explains why large atoms are more reactive than small atoms? a. large atoms have valence electrons farther from the nucleus and lose them more readily. b. large atoms have greater ionization energy, which they can utilize during a reaction. c. large atoms have a greater number of electrons that they can lose during a reaction. d. large atoms have more energy levels, so they have more energy to pass on in a reaction. reset next
Answers: 3

Chemistry, 23.06.2019 02:00
As light moves from one material into the next, which of the following affects how much the light waves will refract, or bend? angle at which the ray strikes the medium color of the material density of the material temperature of the light wave
Answers: 2
You know the right answer?
Which of the following will be more reactive than magnesium ()...
Questions

Mathematics, 04.02.2020 08:44




Mathematics, 04.02.2020 08:44

Mathematics, 04.02.2020 08:44

Computers and Technology, 04.02.2020 08:44

English, 04.02.2020 08:44


Mathematics, 04.02.2020 08:44


Computers and Technology, 04.02.2020 08:44

English, 04.02.2020 08:44





Health, 04.02.2020 08:44

History, 04.02.2020 08:44

Business, 04.02.2020 08:44