
Decide which of the following statements are true and which are false about equilibrium systems: a large value of k means the equilibrium position lies far to the right. for the following reaction: h2(g) + f2(g) ⇌ 2hf(g) the values of k and kp are not the same. the value of k at constant temperature does not depend on the amounts of reactants and products that are mixed together initially. for the following reaction: caco3(s) ⇌ cao(s) + co2(g) the [caco3] appears in the denominator of the equilibrium expression. for a reaction with k > > 1, the rate of the forward reaction is less than the rate of the reverse reaction at equilibrium.

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 06:30
The following reaction shows the products when sulfuric acid and aluminum hydroxide react. al(oh)3 + h2so4 → al2(so4)3 + h2o the table shows the calculated amounts of reactants and products when the reaction was conducted in a laboratory. sulfuric acid aluminum hydroxide initial amount of reactant 40 g 15 g theoretical yield of water from reactant 14.69 g 10.38 g what is the approximate amount of the leftover reactant? 11.73 g of sulfuric acid 10.33 g of sulfuric acid 11.12 g of aluminum hydroxide 13.67 g of aluminum hydroxide
Answers: 3

Chemistry, 22.06.2019 08:30
The characteristic of two different types of reactions are shown below. reaction a: electrons are gained by the atoms of an element. reaction b: protons are lost by the atom of and element. which statement is true about the atoms of the elements that participate in the two reactions? a: their identity changes in both reaction a and b. b: their identity changes in reaction a but not b. c: their identity changes in reaction b but not a. d: their identity remains the same.
Answers: 1

Chemistry, 22.06.2019 13:30
What are the chemical names of these compounds? ke: mg3n2: reset next
Answers: 1
You know the right answer?
Decide which of the following statements are true and which are false about equilibrium systems: a l...
Questions



Mathematics, 31.01.2021 04:30


Biology, 31.01.2021 04:30

History, 31.01.2021 04:30

Mathematics, 31.01.2021 04:30

Mathematics, 31.01.2021 04:30




Business, 31.01.2021 04:30





Chemistry, 31.01.2021 04:30


Mathematics, 31.01.2021 04:30

Mathematics, 31.01.2021 04:30

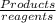
![K= \frac{[C]^{c}[D]^{d}}{[A]^{a}[B]^{b}}](/tpl/images/0292/7295/c008c.png)
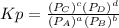
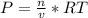 we know that
we know that 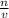 is the molar concentration. When we replace P in the expression for Kp we get:
is the molar concentration. When we replace P in the expression for Kp we get:
![Kp= \frac{[C]^{c}*(RT)^{c}[D]^{d}*(RT)^{d}}{[A]^{a}*(RT)^{a}[B]^{b}*(RT)^{b}}](/tpl/images/0292/7295/8e3a8.png)
![Kp= \frac{[C]^{c}[D]^{d}}{[A]^{a}[B]^{b}}*\frac{(RT)^{c+d}}{(RT)^{a+b}}](/tpl/images/0292/7295/243b8.png)
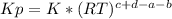
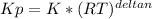
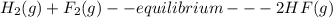
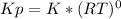
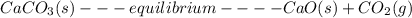
![K= [CO_{2}]](/tpl/images/0292/7295/89ef2.png)
![[CaCO_{3}]](/tpl/images/0292/7295/84a01.png) is not include in the expression.
is not include in the expression. 

