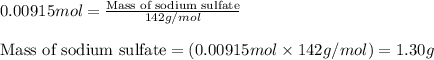Chemistry, 06.10.2019 09:02 eastonstelter
One step in the isolation of pure rhodium metal (rh) is the precipitation of rhodium(iii) hydroxide from a solution containing rhodium(iii) sulfate according to the following balanced chemical equation: rh₂(so₄)₃(aq) + 6naoh(aq) → 2rh(oh)₃(s) + 3na₂so₄(aq) of 0.730 g of rhodium(iii) hydroxide is produced, what mass of sodium sulfate is also produced?

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 14:30
Which is true of the reactants in this displacement reaction? fe + 2hcl fecl2 + h2 a. the reactants are located to the left of the arrow in the chemical equation. b. the reactants contain 1 iron atom, 2 hydrogen atoms, and 1 chlorine atom. c. the reactants are the atoms, molecules, or compounds formed in the reaction. d. the reactants have the same physical and chemical properties as the products.
Answers: 1

Chemistry, 21.06.2019 17:00
Look at the reaction below: ca(hco3)2 --> caco3 + co2 + h2o first, balance the reaction. once balanced, use dimensional analysis or another method to find out how many moles of carbon dioxide will be produced if we start with 16.5 moles of calcium bicarbonate (calcium hydrogen carbonate). = mol of co2 number needs to be reported to three significant figures.
Answers: 1


Chemistry, 22.06.2019 03:30
In this chemical reaction, 325 grams of barium (ba) react completely. how many moles of lithium (li) are produced?
Answers: 1
You know the right answer?
One step in the isolation of pure rhodium metal (rh) is the precipitation of rhodium(iii) hydroxide...
Questions

Mathematics, 11.12.2020 01:00


Biology, 11.12.2020 01:00

Mathematics, 11.12.2020 01:00


Social Studies, 11.12.2020 01:00


Mathematics, 11.12.2020 01:00

Mathematics, 11.12.2020 01:00

Mathematics, 11.12.2020 01:00

Mathematics, 11.12.2020 01:00


Mathematics, 11.12.2020 01:00

English, 11.12.2020 01:00

Mathematics, 11.12.2020 01:00

Mathematics, 11.12.2020 01:00

Mathematics, 11.12.2020 01:00

Mathematics, 11.12.2020 01:00

English, 11.12.2020 01:00

Mathematics, 11.12.2020 01:00

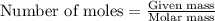 .....(1)
.....(1)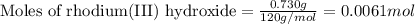
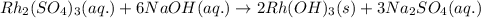
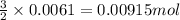 of sodium sulfate is also produced
of sodium sulfate is also produced