
Predict (to 3 sig figs) the isochoric specific heat of gaseous sf6 (molar mass 146.06 g/mol) at 1200 k, assuming that at this temperature all translational, rotational and vibrational degrees of freedom are accessible, and assuming no electronic degrees of freedom are accessible at all. do you need to assume ideal fully gas behavior?

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 23:00
Select the correct answer. given: 2libr + ba → babr2 + 2li in this chemical reaction, 325 grams of barium (ba) react completely. how many moles of lithium (li) are produced? a. 1.18 mol b. 2.37 mol c. 4.73 mol d. 16.4 mol e. 32.9 mol
Answers: 2

Chemistry, 22.06.2019 23:00
What is a substance? a. a physical property of matter b. a chemical property of matter c. an element or compound that cannot be physically separated d. characteristics used to tell the difference between mixtures
Answers: 1

Chemistry, 23.06.2019 00:00
What conclusion can you draw from this experiment about the components of the black ink?
Answers: 3

Chemistry, 23.06.2019 06:10
How can liquids be seperated by density a the liquids are absorbed onto a paper b the liquids are turned into seperate vapors c the liquids are collected as they evaporate d the liquids are allowed to seperate into layers
Answers: 1
You know the right answer?
Predict (to 3 sig figs) the isochoric specific heat of gaseous sf6 (molar mass 146.06 g/mol) at 1200...
Questions

History, 09.10.2019 05:10

English, 09.10.2019 05:10

Mathematics, 09.10.2019 05:10

Biology, 09.10.2019 05:10

History, 09.10.2019 05:10

Mathematics, 09.10.2019 05:10


Business, 09.10.2019 05:10

Chemistry, 09.10.2019 05:10


Mathematics, 09.10.2019 05:10

History, 09.10.2019 05:10



Advanced Placement (AP), 09.10.2019 05:10





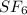 molecule is a non-linear molecule. Therefore, its isochoric heat capacity will be as follows.
molecule is a non-linear molecule. Therefore, its isochoric heat capacity will be as follows.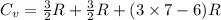
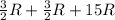
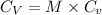
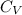 = molar heat capacity
= molar heat capacity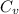 = specific heat
= specific heat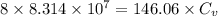

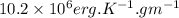 .
.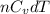
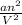 " has to be added here.
" has to be added here.


