Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 00:00
Alarge marble is dropped in a graduated cylinder with 35ml of water in it.the water level increases to 49ml.what is the volume of the marble
Answers: 1

Chemistry, 22.06.2019 03:00
Atrain travels 74 kilometers in 3 hours, and then 81 kilometers in 5 hours. what is its average speed?
Answers: 2

Chemistry, 22.06.2019 05:30
Arecipe calls for 1.2 cups of oil. how many liters of oil is this?
Answers: 2

Chemistry, 22.06.2019 20:20
The characteristics of two different types of reactions are shown below: reaction a: electrons are gained by the atoms of an element. reaction b: protons are lost by the atom of an element. which statement is true about the atoms of the elements that participate in the two reactions? their identity changes in both reaction a and reaction b. their identity changes in reaction a but not in reaction b. their identity changes in reaction b but not in reaction a. their identity remains the same in both reaction a and reaction b.
Answers: 1
You know the right answer?
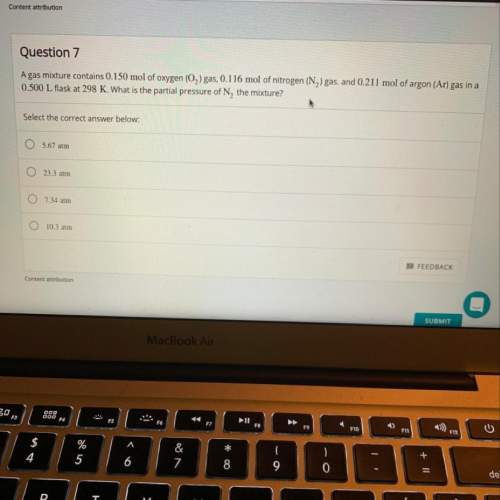
Agas mixture contains 0.150 mol of o2 gas, 0.116 mol of n2 gas, and 0.211 mol of ar gas in a 0.500 l...
Questions


Computers and Technology, 26.11.2020 01:20

Social Studies, 26.11.2020 01:20

Mathematics, 26.11.2020 01:20



Computers and Technology, 26.11.2020 01:20

Mathematics, 26.11.2020 01:20

Arts, 26.11.2020 01:20




Mathematics, 26.11.2020 01:20

Mathematics, 26.11.2020 01:20

Mathematics, 26.11.2020 01:20

History, 26.11.2020 01:20

Mathematics, 26.11.2020 01:20

Mathematics, 26.11.2020 01:20