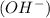13. copper(ii) chloride and silver acetate acid/base:
14. hydrofluoric acid and barium hydro...

Chemistry, 17.10.2019 18:10 daniellealex
13. copper(ii) chloride and silver acetate acid/base:
14. hydrofluoric acid and barium hydroxide
15. magnesium hydroxide and hydrochloric acid
16. lithium hydroxide and acetic acid
17. sulfuric acid and sodium hydroxide
18. sodium carbonate and nitric acid
19. sodium bicarbonate and sulfuric acid
20. zirconium(iv) hydroxide and phosphoric acid
21. sodium sulfite and hydrofluoric acid
22. sodium sulfite and hydrochloric acid
23. sodium sulfide and hydrochloric acid
24. ammonium chloride and sodium hydroxide

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 11:00
Imagine that twenty i.u.’s of enzyme z were catalyzing the above reaction for one minute, under vmaxconditions, in a 3.00 ml assay volume. the assay is buffered with 20 mm phosphate buffer, ph 7.60. what will the ph be at the end of that one minute?
Answers: 2


Chemistry, 22.06.2019 21:00
Write a balanced equation showing the formation of copper (ii) nitrite from its elements
Answers: 1

Chemistry, 22.06.2019 23:30
Match each statement with the state of matter it describes
Answers: 3
You know the right answer?
Questions

Mathematics, 16.10.2020 08:01


Biology, 16.10.2020 08:01



Health, 16.10.2020 08:01

Mathematics, 16.10.2020 08:01



History, 16.10.2020 08:01



History, 16.10.2020 08:01


Mathematics, 16.10.2020 08:01




Mathematics, 16.10.2020 08:01

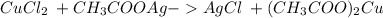
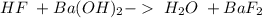
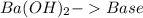
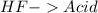

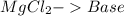
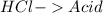
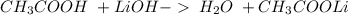
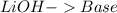
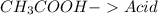
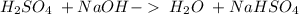

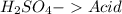
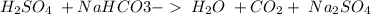
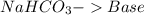
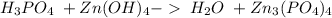
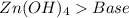
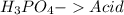
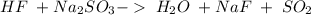
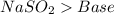
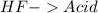
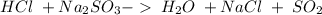
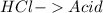
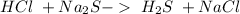
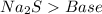
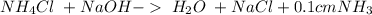
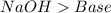
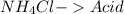
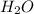 and a salt. Whit this in mind the acids would the compounds that produces the hydronium ion
and a salt. Whit this in mind the acids would the compounds that produces the hydronium ion 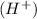 and the bases would be the compounds that produces the hydroxide ion
and the bases would be the compounds that produces the hydroxide ion