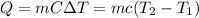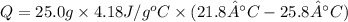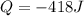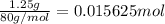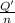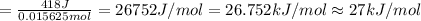Chemistry, 19.10.2019 00:00 tjacqueline9753
In order to measure the enthalpy change for this reaction, 1.25 g of nh4no3 is dissolved in enough water to make 25.0 ml of solution. the initial temperature is 25.8 degrees c and the final temperature (after the solid dissolves) is 21.9 degrees c. calculate the change in enthalpy for the reaction. (use 1.0g/ml as the density of the solution and 4.18 j/g . degrees c as the specific heat capacity.) express the answer to two significant figures.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 09:00
Astudent is asked to identify and element that is pale yellow brittle solid and does not conduct electricity. at which location in this periodic table would the element most likely be found?
Answers: 2

Chemistry, 22.06.2019 10:10
When water dissociates, each water molecule splits into a hydroxide ion and a) h 3 o + b) a hydrogen atom c) a hydrogen ion d) h 2 o e) oh —
Answers: 2

Chemistry, 22.06.2019 15:00
Large helium-filled balloons are used to lift scientific equipment to high altitudes. what is the pressure inside such a balloon if it starts out at sea level with a temperature of 10.0ºc and rises to an altitude where its volume is twenty times the original volume and its temperature is – 50.0ºc ?
Answers: 2

Chemistry, 22.06.2019 19:10
Astudent completes a titration by adding 12.0 milliliters of naoh(aq) of unknown concentration to 16.0 milliliters of 0.15 m hcl(aq). what is the molar concentration of the naoh(aq)? 1)5.0 m 2)0.20 m 3)0.11 m 4)1.1 m
Answers: 1
You know the right answer?
In order to measure the enthalpy change for this reaction, 1.25 g of nh4no3 is dissolved in enough w...
Questions

Chemistry, 24.07.2019 19:00


Computers and Technology, 24.07.2019 19:00

Biology, 24.07.2019 19:00

History, 24.07.2019 19:00

Mathematics, 24.07.2019 19:00



Mathematics, 24.07.2019 19:00

English, 24.07.2019 19:00






Mathematics, 24.07.2019 19:00

Mathematics, 24.07.2019 19:00

Mathematics, 24.07.2019 19:00


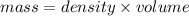
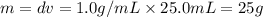
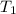 = 25.8°C
= 25.8°C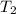 = 21.8°C
= 21.8°C